A path towards the conservation and recovery of Guloninae species worldwide
* This article is part of a Special Issue from the Martes Working Group.
1Ministry of Water, Land and Resource Stewardship, Victoria, British Columbia, Canada
2Department of Natural Resource Sciences, Thompson Rivers University, Kamloops, British Columbia, Canada
3Oregon State University, Corvallis, Oregon, USA
4Department of Forestry and Wildlife Management, Inland Norway University of Applied Sciences, Koppang, Norway
5Institut de Recherche sur Les Forêts, Université du Québec en Abitibi-Témiscamingue, Rouyn-Noranda, Québec, Canada
6College of Forest Resources and Environmental Science, Michigan Technological University, Houghton, Michigan,USA
7British Small Animal Veterinary Association, Woodrow House, Waterwells Business Park, Quedgeley, UK
8School of Environmental Studies, University of Victoria, Victoria, British Columbia, Canada
9Department of Biology, Central Connecticut State University, New Britain, Connecticut, USA
10Department of Wildlife, Fish, and Environmental Studies, Swedish University of Agricultural Sciences, Umeå, Sweden
11Olympic National Forest, United States Department of Agriculture Forest Service, Washington, USA
12Gloucestershire Wildlife Trust, Coleford, England, UK; Royal Society for the Protection of Birds (RSPB), Somerset, England, UK
13Vincent Wildlife Trust, Ledbury, Herefordshire, UK
14Fondazione Ethoikos, Convento dell’Osservanza S.N.C., Radicondoli, Siena, Italy
15Ural Branch of the Russian Academy of Sciences, Institute of Plant and Animal Ecology,Yekaterinburg, Russia
16Coordination Mammalogique du Nord de la France, Vimy, France
17Institute of Zoology, Almaty, Kazakhstan
18Forest Research, Alice Holt Research Station, Farnham, UK
19 WSP UK Limited, Edinburgh, Scotland, UK
20 Conservation Biology Institute, Corvallis, Oregon, USA
JMB: https://orcid.org/0000-0003-4724-5286
SY: https://orcid.org/0009-0004-9698-3781
EA: https://orcid.org/0000-0002-6846-2928
SB: https://orcid.org/0000-0002-9337-8234
NC: https://orcid.org/0009-0007-9824-5442
SRC: https://orcid.org/0000-0002-6465-5546
EC: https://orcid.org/0000-0001-6353-4550
JTF: https://orcid.org/0000-0002-9020-6509
IJH: https://orcid.org/0009-0002-4034-1662
PH: https://orcid.org/0000-0002-1171-2615
TRH: https://orcid.org/0000-0003-2101-5482
JK: https://orcid.org/0009-0000-9650-098X
JM: https://orcid.org/0000-0002-6919-120X
EM: https://orcid.org/0000-0001-8861-1559
VCM: https://orcid.org/0000-0003-3021-0116
CP: https://orcid.org/0009-0000-3063-0095
AYO: https://orcid.org/0000-0003-3079-8036
ERS: https://orcid.org/0000-0002-4410-5985
HS: https://orcid.org/0009-0002-1343-1275
WDS: https://orcid.org/0009-0003-9859-1872
Burgar, J. M., Yaeger, S., Anderson, E., Brainerd, S., Chabaud, N., Cotey, S. R., Croose, E., Fisher, J. T., Hansen, I. J., Hapeman, P., Hofmeester, T. R., Howell, B. L., Kingscott, J., MacPherson, J., Manzo, E., Monakhov, V. G., Poirson, C., Oleynikov, A. Y., Scopes, E. R., Smith, H., & Spencer, W. D. (2025). A path towards the conservation and recovery of Guloninae species worldwide. Stacks Journal: 25007. https://doi.org/10.60102/stacks-25007
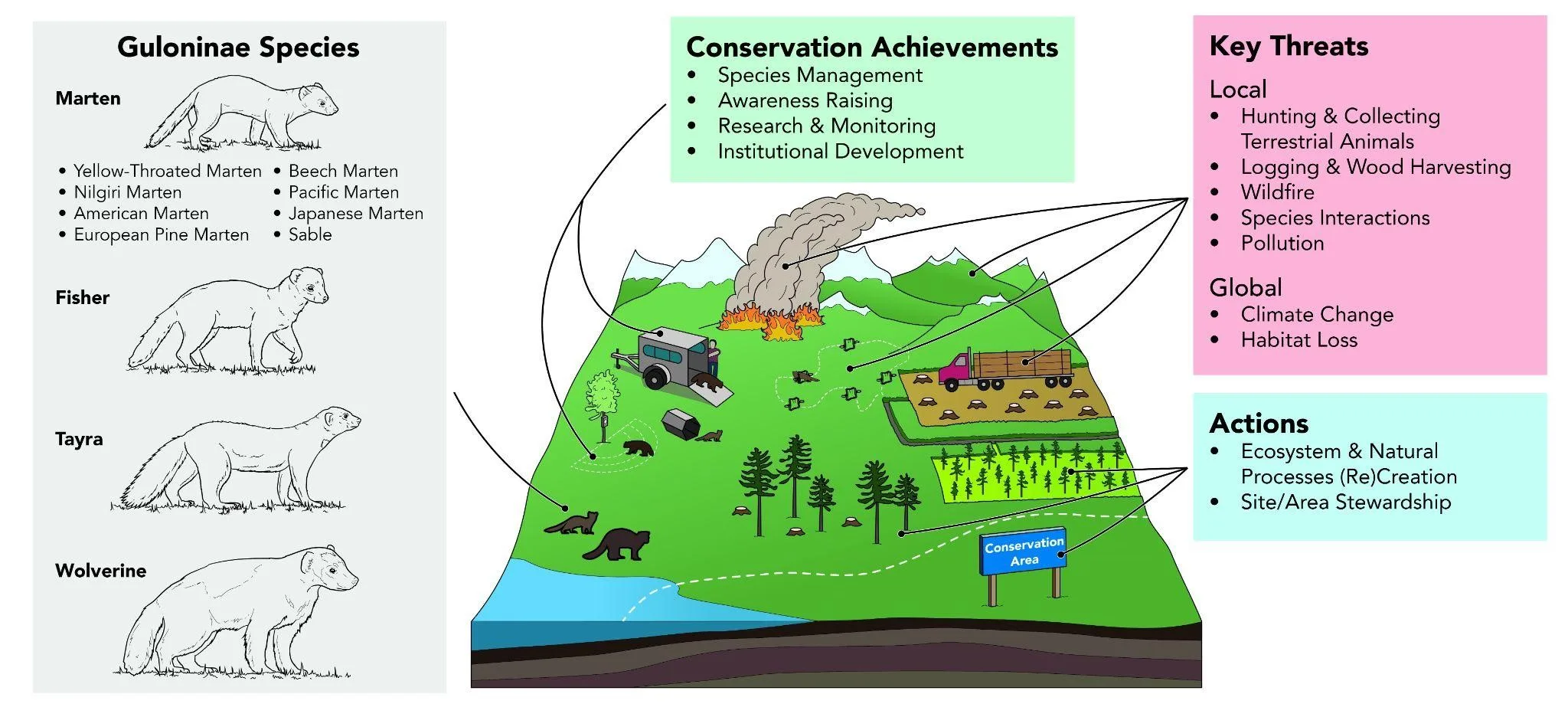
Abstract photo. Global conservation achievements, key threats, and actions necessary to further Guloninae conservation and recovery, as identified by Martes Working Group members and symposium participants as a 3-phase exercise in combining expert opinion information.
Abstract
Global biodiversity is declining as human impacts increase and mammals, especially carnivores, are declining particularly quickly. Small carnivores (such as in the Guloninae subfamily) are less frequently considered than large carnivores, despite also being affected by biodiversity declines, because their status and population ecology are exceedingly difficult to assess. Although all but one Guloninae species are globally listed as Least Concern by the International Union for Conservation of Nature (IUCN), this designation obscures conservation challenges and does not include local population declines. Proactive efforts to improve population persistence in regions of most concern may prevent species from becoming globally threatened in the future. We briefly introduce the 11 Guloninae species, highlight collective challenges, and synthesize possible options focused on recovery and conservation. To inform our paper, we conducted a 3-phase process to gather expert opinions: an online survey, an in-person prioritization exercise of online results, and directed in-person group discussions. We focused efforts within the international Martes Working Group consisting of species experts. We followed the IUCN and Conservation Measures Partnership (CMP) threats and actions classifications nomenclature to enable cross-project learning and data aggregation. From our results, the largest described global threat to Guloninae was ‘Climate Change’ (e.g., ecosystem encroachment; changes in temperature regimes) as most Guloninae species appear to have a narrow thermal range where persistence is possible. The largest threat at the jurisdictional level was ‘Biological Resource Use’ (e.g., hunting and collecting terrestrial animals; logging and wood harvesting). The identified action needed to further Guloninae conservation and recovery was ‘Land/Water Management’ (e.g., ecosystem and natural process re-creation; site/area stewardship). We identified unifying themes threatening and benefitting Guloninae; but given the wide geographic range of Guloninae species, the many countries involved, and the diversity of ecosystems with different levels of human impacts, conservation actions need to be locally appropriate. Our paper synthesizes natural history and guidance from experts to provide a foundation for future research efforts and conservation actions for Guloninae conservation.
Keywords: conservation actions, fisher, marten, mustelids, Qualitative analysis, tayra, thematic coding, threats, wolverine
Introduction
Global biodiversity is declining as the human footprint increases (Maxwell et al. 2016) and populations of large mammals, especially terrestrial carnivores, have experienced historical and ongoing declines (Ingeman et al. 2022). Small carnivores (i.e., members of the order Carnivora with an average body mass <21.5 kg; Do Linh San et al. 2022) are also impacted, though the status of these species and their population ecology are much less known (Marneweck et al. 2021). Many of these species hold important cultural significance to Indigenous Peoples (Moore and Wheelock 1990; Badmaev 2003; Gura 1997) and they can be of economic significance (Schreiber et al. 1989). However, small carnivores can be considered pest or nuisance species (Herr et al. 2010; Peeva and Raichev 2016; Swenson and Andrén 2005) as they often interact or inhabit spaces with humans (Mallick 2015; Basnet and Rai 2020; Ballejo et al. 2022; Shameer et al. 2023) and can occupy areas close to urbanized landscapes (Streicher et al. 2023). These small carnivores are often characterized by declining populations (Fogarty et al. 2022; Grattarola et al. 2024) and they face unique conservation challenges.
Small carnivores in the subfamily Guloninae (Gray 1825), within the family Mustelidae, include active predators, omnivores, scavengers, and seed dispersers (Bonaccorso et al.1980; Camargo-Sanabria and Mendoza 2016; Cui et al. 2023; Tsuji et al. 2020; Pereira et al. 2019). These animals can maintain important ecosystem roles due to their intermediate trophic level between primary and tertiary consumers (Marneweck et al. 2022). Due to the extirpation of many large carnivorans, some Guloninae species have become the dominant carnivorans in their ecosystem, shifting their ecological role (Chiang et al. 2012; Kang et al. 2023). Understanding the unique and changing ecology of these species, and threats to their persistence, are prerequisites to establishing effective conservation decisions.
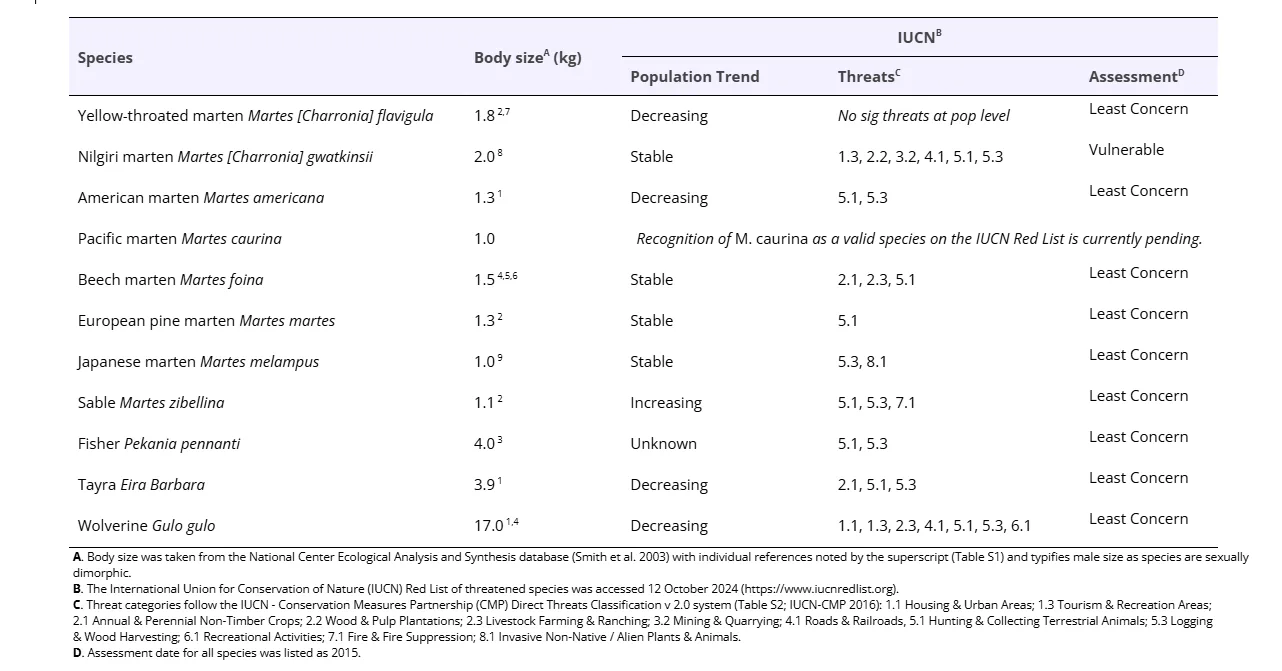
Guloninae species occur throughout Asia, North and South America, and Europe (Figure 1), have many ecological similarities, and face similar threats (Table 1). Although most Guloninae species are globally listed as Least Concern by the International Union for Conservation of Nature (IUCN; Table 1), there have been significant local declines for many populations. Acting now to improve population persistence may prevent these species from becoming globally threatened in the future. Finding commonalities can improve our abilities to conserve local populations, especially given the unequal research and attention paid to individual species and geographic regions.
In this paper we first briefly introduce each of the 11 Guloninae species. We then highlight the conservation achievements, threats, and actions, as identified by Guloninae species experts during a 3-phase process (i.e., online survey, an in-person prioritization exercise of online results, and directed in-person group discussions). Lastly, we synthesize possible steps towards Guloninae recovery and conservation, leveraging lessons learned for individual species more broadly.
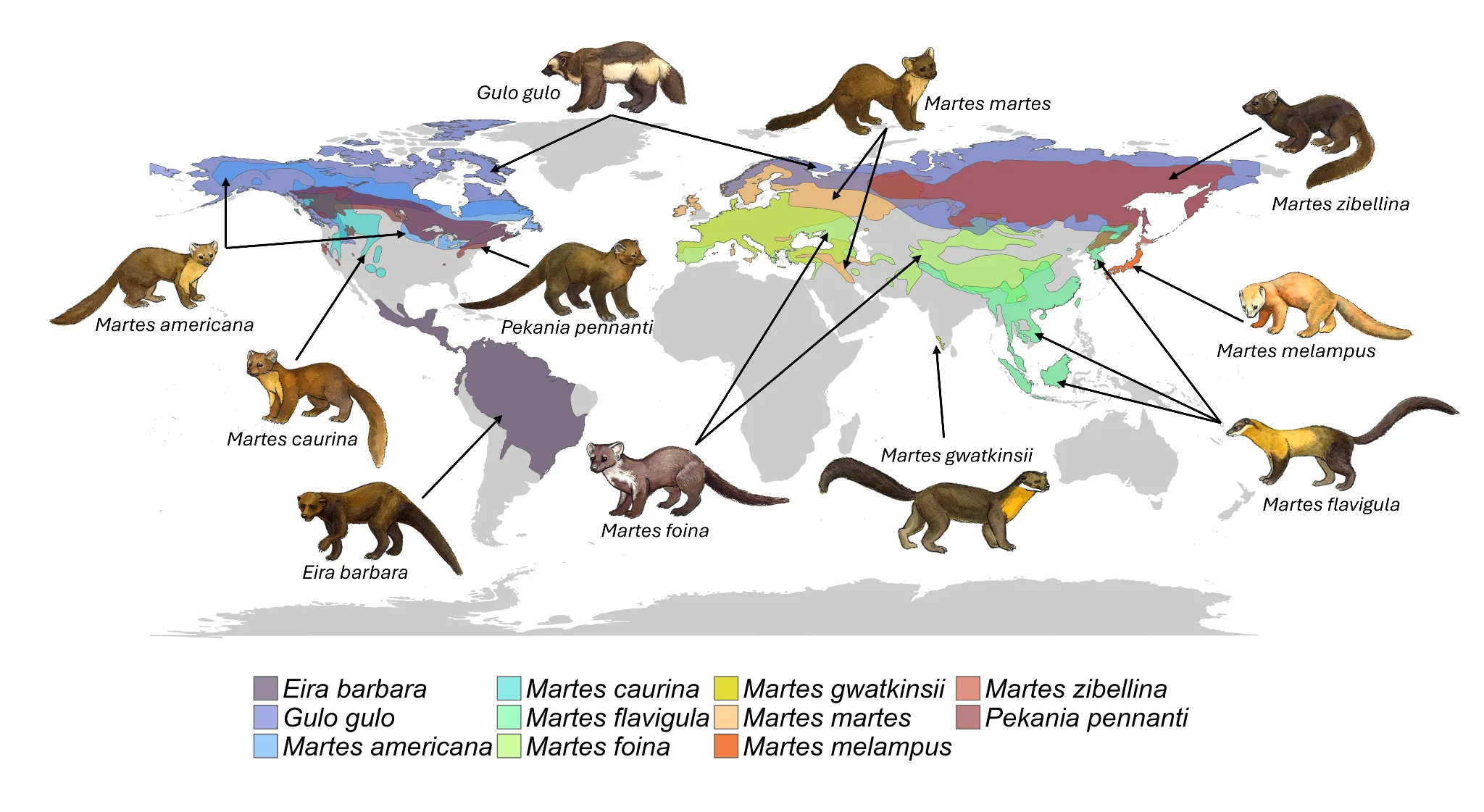
Figure 1. The global ranges of Guloninae species. Range data for most species was downloaded from the International Union for the Conservation of Nature (IUCN), with the exception of Martes americana and M. caurina where range data was extracted from the Mammal Diversity Database (Fernandez 2024). The artist (E. Scopes) also made slight changes. Ranges of several species (and corresponding colors on this map) overlap. We acknowledge that range maps are ever evolving and recommend that readers look to the Martes Working Group website for the most up to date range maps.
Species profiles

Yellow-throated marten (Martes [Charronia] flavigula)
The yellow-throated marten (of the subgenus Charronia) occupies a diverse range of habitats across Asia, spanning from temperate broadleaf and mixed forests (i.e., coniferous-deciduous) to tropical rainforests (Pocock 1941; Heptner and Naumov 1998). The species’ range is fragmented on the mainland, but includes large islands (e.g., Taiwan, Hainan, Borneo, Sumatra, and Java; Chutipong et al. 2016). They often den in tree hollows and are closely associated with old-growth, high-canopy forests (Parr and Duckworth 2007; Oleynikov et al. 2022; Yudin and Yudina 2022).
Conservation status of the yellow-throated marten varies by country; the most vulnerable populations are located in the northern and western parts of the species’ range (e.g., Russia, China, North Korea, South Korea, Taiwan, India), while they are more common in Southeast Asia (Parr and Duckworth 04 2007; Chutipong et al. 2016). Major threats include deforestation, forest degradation, and habitat fragmentation (Chutipong et al. 2016). Poaching (e.g., hunting for fur, mortality due to hunting dogs, accidental deaths in traps, and snares intended for other species) remains a persistent threat (Basnet and Rai 2020; Oleynikov et al. 2022). In some areas, road mortality (Kang et al. 2023) and mortality due to human-wildlife conflicts are problematic (Mallick 2015). Yellow-throated martens can live in areas of high population density (e.g., Lee et al. 2021, Kang et al. 2023), so understanding population risks may be additionally helpful in these areas.

Nilgiri marten (Martes [Charronia] gwatkinsii)
The Nilgiri marten (of the subgenus Charronia) inhabits the southwestern region of India in the Western Ghats, and has the smallest range of all Guloninae species. The range is fragmented into five distinct patches, occupying the highest elevation montane areas with well-preserved forests (Shameer et al. 2023). The Nilgiri marten is primarily found in evergreen forests and montane forest-grassland mosaics, with occasional sightings in moist deciduous forests near evergreen forests (Mudappa et al. 2015; Kumar and Yoganand 1999). Major threats include deforestation and habitat fragmentation due to road construction and dam development, as well as poaching and persecution by beekeepers in retaliation for raiding bee-boxes (Mudappa et al. 2015; Shameer et al. 2023). The Nilgiri marten has received the least amount of research focus of all Guloninae species.

American marten (Martes americana)
American martens are found in North America and their distribution is thought to be a function of a northward population expansion during glacial retreat and expanding boreal forest conditions (Dawson and Cook 2012). American martens occur east of the Rocky mountain crest and north into the Great Lakes region, the northeastern United States, and across Canada from Newfoundland to British Columbia (Wright 1953; Dawson and Cook 2012; O’Brien et al. 2018, Colella et al. 2024). This species is associated with late-successional, mesic coniferous, and mixed-hardwood forests (Powell et al. 2003; Ashbrook and Hapeman 2024).
Contemporary conservation concerns for American martens include interspecific competition, climate change (Manlick et al. 2020), and forest harvesting (Woollard et al. 2024). American martens and fishers both have overlapping ranges and diets (Manlick et al. 2017; Smith et al. 2023) and are competitors that nonetheless coexist throughout their range, a balance that is believed to be maintained by differential responses to topography, climate events, and landscape change (Pauli et al. 2022). However, co-existence may be reduced in human-dominated landscapes, where American martens are not as well-equipped as fishers to inhabit these novel ecosystems (Manlick et al. 2020). Old, mixed, and coniferous forests are associated with habitat for American martens (Powell et al. 2003; Cheveau et al. 2013), forest loss and fragmentation is reducing conifer composition (Danneyrolles et al. 2016). Reduction in snow cover and formation of a crust on snow cover, linked to climate change (Thompson et al. 2021), could reduce the quality and accessibility of the subnivium (Suffice et al. 2020; Pauli et al. 2022), a thermal refuge zone where martens can forage efficiently (Pauli et al. 2013, 2022).

Pacific marten (Martes caurina)
Pacific martens are a distinct species from American marten (Merriam 1890; Wright 1953; Dawson and Cook 2012). They are distributed from the west coast of North America into the Rocky Mountains (Dawson et al. 2017, Colella et al. 2018, 2024). Contact zones between American and Pacific martens occur along the central Rocky Mountains of Idaho and Montana and potentially into British Columbia, with an isolated contact zone on Kuiu Island in southeast Alaska (Small et al. 2003, Colella et al. 2024). Similar to American martens, Pacific martens are typically associated with structural complexity (Delheimer et al. 2023) and late-successional forests (Buskirk and Powell 1994; Thompson 2012), but have considerable plasticity including occurring in island ecosystems (Breault et al. 2021) and within coastal dune and interior coastal forests (Moriarty et al. 2021).
Contemporary threats to the persistence of Pacific martens include climate change effects such as increasingly severe wildfires and decreased snowpack reducing available habitat at high elevations (Moriarty et al. 2015; Moriarty et al. 2019; Spencer et al. 2015; Colella et al. 2024); reductions in forest complexity and connectivity brought by fire and other resource management actions (e.g., Moriarty et al. 2015; Martin et al. 2020); and occurrence in small, isolated populations that may be susceptible to stochastic events (e.g., Schwartz et al. 2020; Spencer et al. 2015). Pacific martens may be considerably more sensitive to warming conditions compared to American martens (Colella et al. 2024).

European pine marten (Martes martes)
European pine martens are widespread and exist in forest and scrub environments across Europe, Asia Minor, and have recently expanded into the Irtysh and Ob river basins of Kazakhstan and West Siberia (Herrero et al. 2016; Oleynikov and Monakhov, 2023; Caryl et al. 2012; Lombardini et al. 2015; Monakhov 2022). European pine martens are dietary generalists (Helldin 1999, 2000). They can use a wide variety of forest types, but tend to avoid open areas such as bogs and clearcuts, preferring mature stands (Brainerd and Rolstad 2002).
Legal protections for the European pine marten vary by jurisdiction. They are legally hunted and trapped in many parts of their range (e.g., Austria, France, Norway, and Sweden). Although they were extirpated in many parts of Europe due to overharvest, they have recently begun to recover some of their former range with the help of full legal protection and conservation translocations (Balestrieri et al. 2009; De Groot et al. 2016; Helldin 2000; Herrero et al. 2016; Manzo et al. 2018; McNicol et al. 2020; Proulx et al. 2004; Van Den Berge et al. 2000). After longer periods of protection, European pine marten populations gradually increased, and fur harvest records peaked concurrently with a drastic decline in red fox (Vulpes vulpes) populations due to sarcoptic mange (Lindström et al. 1995). Harvest records indicate that European pine marten populations have declined subsequent to the recovery of the red fox populations in the 1990s and may be sensitive to local overharvest (Helldin 2000). European pine martens are also threatened by habitat loss, persecution, loss of population connectivity due to road networks (Mathews et al. 2018; Monakhov 2022; Genovesi et al. 2015), and illegal killings relating to the protection of farmed and game poultry (Sainsbury et al. 2019).

Japanese marten (Martes melampus)
The Japanese marten is endemic to the forests of Honshu, Shikoku, and Kyushu islands, and was introduced to Hokkaido and Sado Island (Murakami and Ohtaishi 2000). They can be found in lowlands and mountains, in both deciduous and evergreen forests (Masuda 2009), but show a preference for broadleaved forests (Tatara 1994; Hoshino et al. 2014). They are dietary generalists (Tsuji et al. 2019) and important long distance seed dispersers, particularly in sub-alpine shrubland (Otani 2002).
Most of the Japanese marten population is stable and subject to hunting regulations. However, some of the local populations require conservation efforts (Proulx et al. 2004; Masuda 2009). Japanese marten on Gunma Prefecture, Aichi Prefecture, and Tsushima Island are experiencing habitat loss and degradation from intensive deforestation, plantation establishment, and urbanization (Tatara and Doi 1994; Proulx et al. 2004; Abramov et al. 2015; Hisano et al. 2018), and have been placed on Japanese federal and regional Red Lists. Habitat destruction and degradation exacerbate the isolation of distinct population clusters. Additionally, the introduction of invasive mustelids (i.e., American mink Neogale vison and Siberian weasel Mustela sibirica) are recognized as a mortality threat through competition (Abramov et al. 2015). The Tsushima population (M. m. tsuensis) exhibits lower genetic variation compared to populations in the Japanese archipelago, suggesting a higher conservation priority (Sato et al. 2009).

Sable (Martes zibellina)
Sables are widespread and inhabit lowland and mountain taiga as well as coniferous and deciduous forests in six countries: China, Japan (Hokkaido), Kazakhstan, DPR Korea, Mongolia, and Russia. Most sables are concentrated in Russia (Monakhov and Li 2013), and are well-studied because they are a valuable fur-bearing animal (Bakeev and Sinitsyn 1994; Monakhov 2011, 2016; Proulx et al. 2004). Sables are bred on fur farms in the Russian Federation and are common in zoos in many large cities around the world. Sables are legally hunted and over >160,000 wild sable pelts were offered at 226 international fur auctions in May 2024 (Sojuzpushnina 2024). There has been no evaluation providing evidence of overharvesting in recent decades, and populations of sable appear to be increasing, according to the most recent assessment in 2015 (Monakhov et al. 2016).

Fisher (Pekania pennanti)
Fishers are endemic to North America, and occupy mesic coniferous, conifer-hardwood, and mixed-hardwood forests, extending from the boreal and mixed-transitional forests of Canada and the United States into the mountainous regions of the western United States. The distribution of fishers contracted significantly between the mid-1800s and mid-1900s due to unsustainable trapping for fur, predator- and pest-control campaigns, habitat loss and fragmentation, and climate changes during the Little Ice Age (Douglas and Strickland 1987; Powell and Thompson 1993; Ruggiero et al. 1994; Knaus et al. 2011; Krohner et al. 2022; Tucker et al. 2014; Lofroth et al. 2023). Fishers now reoccupy some portions of their historical range through reintroductions by resource management agencies and natural range expansion (Facka and Powell 2021; Green et al. 2022; Happe et al. 2020; Happeman et al. 2011; Lewis et al. 2022; Stewart et al. 2017).
Fisher recovery in the western portions of their distribution is a conservation concern. Contemporary threats include habitat fragmentation from agriculture (Stewart et al. 2019), logging and wood harvesting (Naney et al. 2012), petroleum exploration and extraction (Fisher et al. 2013; Fisher and Burton 2018), uncharacteristically severe wildfires (Steel et al. 2015; Green et al. 2022; Williams et al. 2023), insect and drought-induced tree mortality (Steventon and Daust 2009), rodenticide poisoning associated with public land cannabis cultivation (Gabriel et al. 2015), fur trapping (Fogarty et al. 2025), and the synergistic effects of these threats on small populations (Naney et al. 2012). While decreases in fisher populations have been observed in some regions in western North America (e.g., Fogarty et al. 2022), their numbers have expanded in eastern North America into more urbanized landscapes, and little is currently understood regarding the differences in behavior and population trajectories across their range (but see LaPoint et al. 2013; Lofroth et al. 2010; Loughry et al. 2012; Olson et al. 2024).

Tayra (Eira barbara)
The tayra is found in neotropical forested habitats from southern Mexico to northern Argentina (Presley 2000; Schiaffini et al. 2017). Although up to 16 subspecies have been previously identified, Schiaffini (2020) was unable to separate subspecies based on the criteria of geographic isolation, ability to interbreed, and morphological and genetic differences. Denning occurs in tree cavities or in burrows within tree root structures and den sites may be a limiting factor in unprotected areas (Bianchi et al. 2021). Tayras prey upon a variety of arboreal and terrestrial animals from insects to medium and large-sized mammals (Grotta-Neto et al. 2021). They also consume and cache fruits, possibly contributing to seed dispersal (Galef et al. 1976; Soley and Alvarado-Díaz 2011). The Mexican government lists the tayra as endangered due to habitat fragmentation and habitat loss (Chavez 2014). Many aspects of tayra ecology are still unknown, limiting conservation efforts throughout its range (Bianchi et al. 2021).

Wolverine (Gulo gulo)
Wolverines have a circumpolar distribution, located mainly in the boreal and arctic regions of the northern hemisphere (Copeland and Kucera 1997). Historically, their range and habitat use were much more extensive, but human development and infrastructure (Fisher et al. 2013; Heim et al. 2017), trapping (Krebs et al. 2004; Scrafford et al. 2024), and predator removal efforts in the early 1900’s reduced their southern range (Copeland and Kucera 1997; May 2007; Walker et al. 2001).
A global analysis suggested the primary threat to wolverines is climate and landscape change (Fisher et al. 2022). Spring snow cover can have important implications for natal den success (Copeland et al. 2010; Barrueto et al. 2022; Magoun and Copeland 1998), and warming temperatures associated with climate change will not only affect natal denning success but also decrease the availability of habitat, particularly in summer months, and further reduce the wolverine’s range (Copeland et al. 2010). Wolverines have very large home ranges (e.g., 1017 km2 for females and 1996 km2 for males; Bischof et al. 2020), which makes them susceptible to the cumulative effects of habitat fragmentation. Protected areas remain important for wolverine persistence, in addition to land and population management in landscapes where they remain (Fisher et al. 2022).
Methods and Materials
Data collection
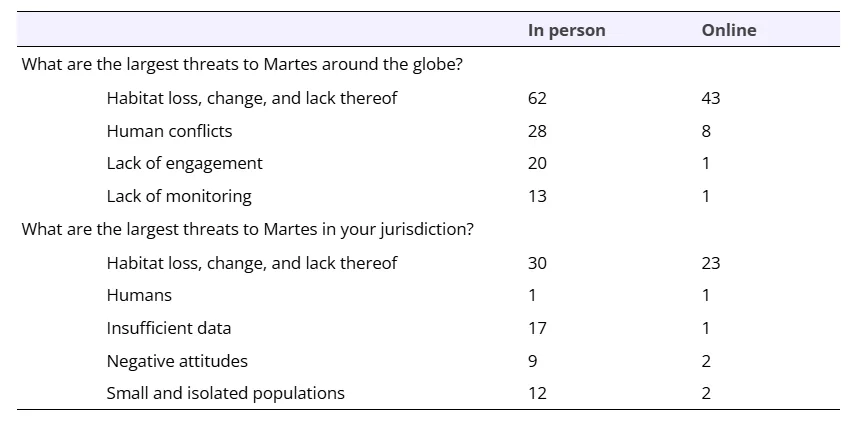
Since 1994, a dedicated group of researchers, scientists, and land managers collectively discuss the state of science every 4-5 years for the 11 Guloninae species (i.e., the “Martes Working Group”), often writing a book following the symposium. Uniquely in 2023, this group participated in a 3-part exercise with the goal of identifying paths for species persistence and recovery: 1) an anonymous online survey, 2) in-person voting, and 3) in-person discussion. We sent an anonymous online survey to all Martes Working Group members via an email and newsletter; the survey was open for one week (August 24 – September 1, 2023). We asked nine questions, including six questions to ascertain respondents’ reflections on conservation achievements, threats, and obstacles for their study species at a local and global scale (Figure S1). Responses to these six questions were then grouped into categories derived from the data (i.e., inductive coding).
The six questions, and categorized responses were posted for review and prioritization over three days at the 8th International Martes Symposium of the Martes Working Group (Aviemore, Scotland, September 12-15, 2023; https://www.martesworkinggroup.org/martes-symposium-2023/). Prioritization consisted of each symposium attendee identifying the categorized response(s) that resonated most to them; attendees affixed up to 18 stickers to as many, or as few, responses as they deemed relevant (Figure 2). Attendees could affix stickers to responses for one, or all, questions. As such, not all attendees may have prioritized categorized responses for each question. Attendees were also given the opportunity to add additional responses if they felt a category was missing. Four additional categories were added by symposium attendees, two each for the questions asking about recent conservation achievements and needed conservation actions.
On the final day of the symposium we collated in-person ranking by counting the number of stickers associated with each categorized response, to determine the highest priority discussion topics (Figure S2). We used the topics that emerged through the online survey and in-person voting to guide the discussion. The intent of the in-person discussion was to enhance knowledge sharing between Guloninae researchers, particularly to ensure representation beyond Europe and North America, and local stakeholders, and creative thinking to secure funding to support representative knowledge sharing. In groups of 5-10 people (n = 12 tables), each group discussed five questions:
- Why do you think these themes emerged?
- How can we overcome obstacles that exist for conservation of Guloninae?
- How can conservationists working with Guloninae species replicate and scale wins?
- How can research best advance conservation and management efforts?
- What actions can we take in the next year or two to lead to more success over the next 5-10 years?
Groups were self-selected and one group member was designated as scribe, taking notes of the discussion. Each table contributed one set of responses per discussion question.
Our research involved human subjects but was exempt from Institutional Review Board review as it primarily involved the collection of expert opinions in a professional setting. The group discussion and survey were conducted in a manner consistent with research on public behavior, where no intervention occurred, and no private, sensitive, or personally identifiable data was collected beyond professional perspectives. Participants voluntarily contributed their expertise in a setting where professional discourse is expected.

Figure 2. A participant at the 8th International Martes Symposium, standing next to white chart paper with recent conservation achievements and needed actions for Guloninae species displayed. Participants are prioritizing threats and themes for our third activity of directed discussion. The participant gave permission to be photographed.
Data coding (Thematic analysis)
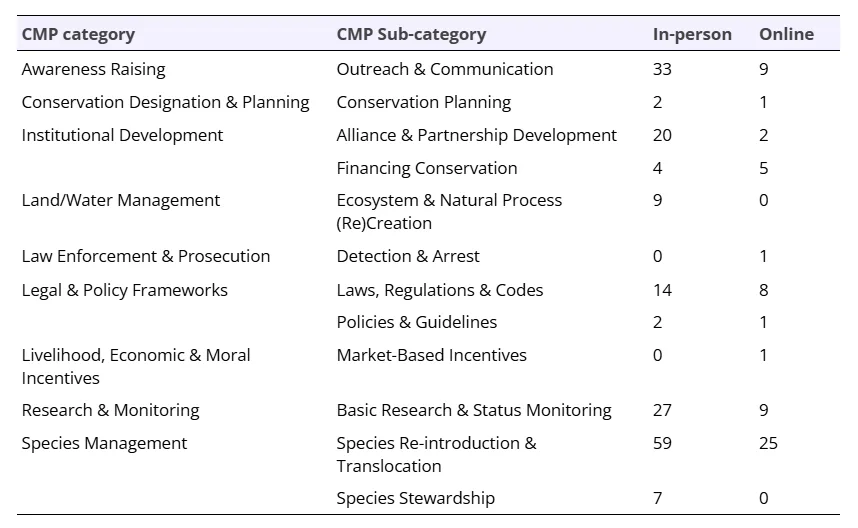
We used content (thematic) analysis to categorize responses to the five questions concerning conservation achievements, threats, and actions. Thematic analysis identifies themes within the data. For the categorization of responses presented to symposium attendees (i.e., in-person ranking and discussion) we followed inductive coding, where the categories were based on the data. We used deductive coding to categorize responses for analysis; deductive coding uses pre-defined codes and assigns them to the qualitative data. We shifted to deductive coding to make the synthesis more broadly applicable to conservation recovery for Guloninae species and beyond. We classified the responses to match the Direct Threats and Action Classifications defined by the Conservation Measures Partnership (CMP) and in conjunction with IUCN (versions 2.0, updated from Salafsky et al. 2008; see Supporting Information). Using an international standard, such as the IUCN categories, allows for comparison with existing, and future, conservation assessments. For consistency, we re-coded the online survey responses, and the symposium responses, to the CMP categories and sub-categories. We calculated the frequency of codes and sub-codes, summing the number of times each sub-category and category response was provided (online survey, in-person discussion) or prioritized (in-person prioritization with stickers). To relate the online survey and in-person prioritization we provide the proportion of responses per number of potential respondents (i.e., number of online survey respondents, n = 46; and number of symposium attendees, n = 101). We provide the percentage of votes graphically, and the count of votes tabularly, highlighting key examples.
Below, we provide an example to illustrate the re-coding process. One original (inductive) category response to the online survey question of ‘What are some conservation/recovery wins for the Martes species you study over the past few years?’ was ‘Species recoveries and recolonizations’. We re-coded this to fit the CMP classification of ‘Species Re-introduction & Translocation’ under the broader classification of ‘Species Management’. Online comments for this coding comprised 15 individual responses, resulting in an online survey count of 15:
- The recovery of the Newfoundland marten,
- Evidence of population expansion,
- Range expansion in SW Scotland,
- Massive recolonisation of Scotland range now moving south into England,
- In Scotland, over the last few decades, the pine marten (Martes martes) has recovered spectacularly, due to a reduction in persecution and a priori expansion in forest cover. In recent years, this expansion has continued, such that martens will soon colonise England by natural means,
Pine martens are recovering in Wales and England, - Pine martens are recovering in Ireland and the UK and are generally being warmly welcomed,
- Increased range and population size through natural recovery,
- Fishers are doing well following reintroduction several decades ago,
- Martens are still rare in WI, but a healthy population has been discovered on the Apostle Islands, possibly a refugia,
- Pine marten recovery aids red squirrel conservation through grey squirrel control and suppression,
- Tracing on expansion to new lands,
- Proven expansion of population where martens have become absent at the hand of man,
Increased detections of marten though animals still seem very rare and at low densities.
The above example highlights the introduced bias and subjectivity of the coder, as original responses may be argued to fit within a separate sub-category (e.g., species stewardship). Coding was conducted by a single author for consistency.
Results
We received 46 responses to our online survey (31% response rate). Most of the respondents were affiliated with universities or colleges (38%), with less representation from non-profit organizations (30%), government agencies (17%), and other groups (15%: retirees = 4, private researchers or consultants = 2 and one tribal organization). Two respondents were associated with both universities or colleges and government agencies and one respondent did not provide an affiliation. Respondents studied Guloninae species around the world, but were primarily focused on species in Europe (49%) and North America (45%), with limited representation in Asia (6%) and South America (2%). One respondent selected multiple continents of focus, whereas all other respondents selected only one continent. Most respondents studied at least one of the eight marten species (85%), followed by fisher (40%), wolverine (17%) and tayra (2%). Percentages do not add to 100 because 15 respondents studied two species (or species groups in the case of marten) and four respondents identified four species / species groups.
Not all Martes Working Group members attended the symposium and not all symposium attendees were Martes Working Group members. Of the Martes Working Group Symposium participants (n = 101), most were affiliated with universities or colleges (35%), followed nearly equally by non-profit organizations (25%), government agencies (22%), and a small number of industry (11%) and other (6%) groups. The majority (68%) of attendees were from Europe, with 29% from North America and only 3 (i.e., 3%) individuals were from Asia or South America. Affiliations and geographic area of study were gleaned from registration and not all attendees entered this information (n=63 and n=97, respectively). Nearly a quarter of attendees were students (23%).
Some original responses could not be coded to CMP categories or sub-categories. This occurred for two questions and was generally due to the vague nature of the original response (Table 2). Removing these responses resulted in 17% fewer online responses and 16% fewer in-person survey responses.
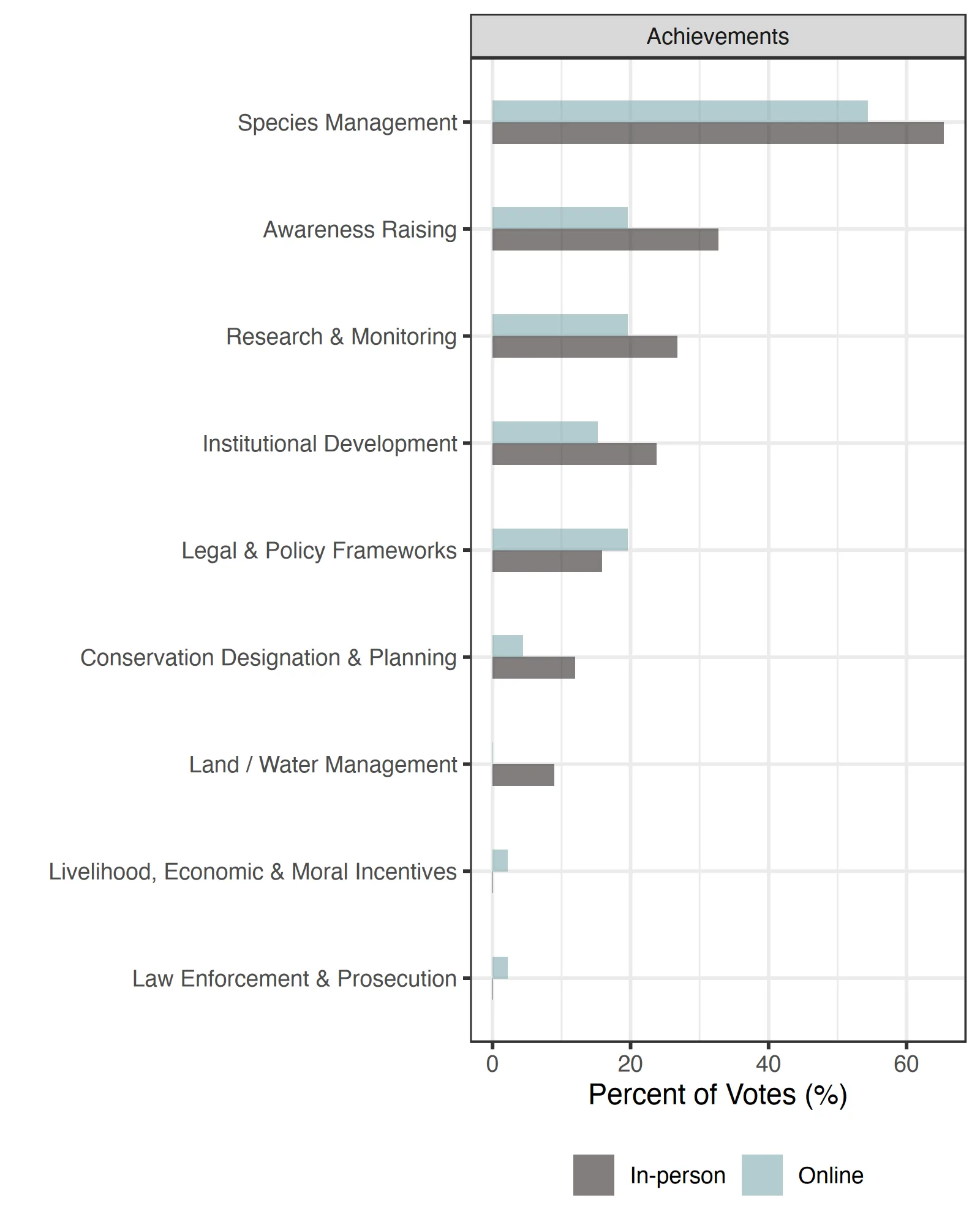
Conservation achievements
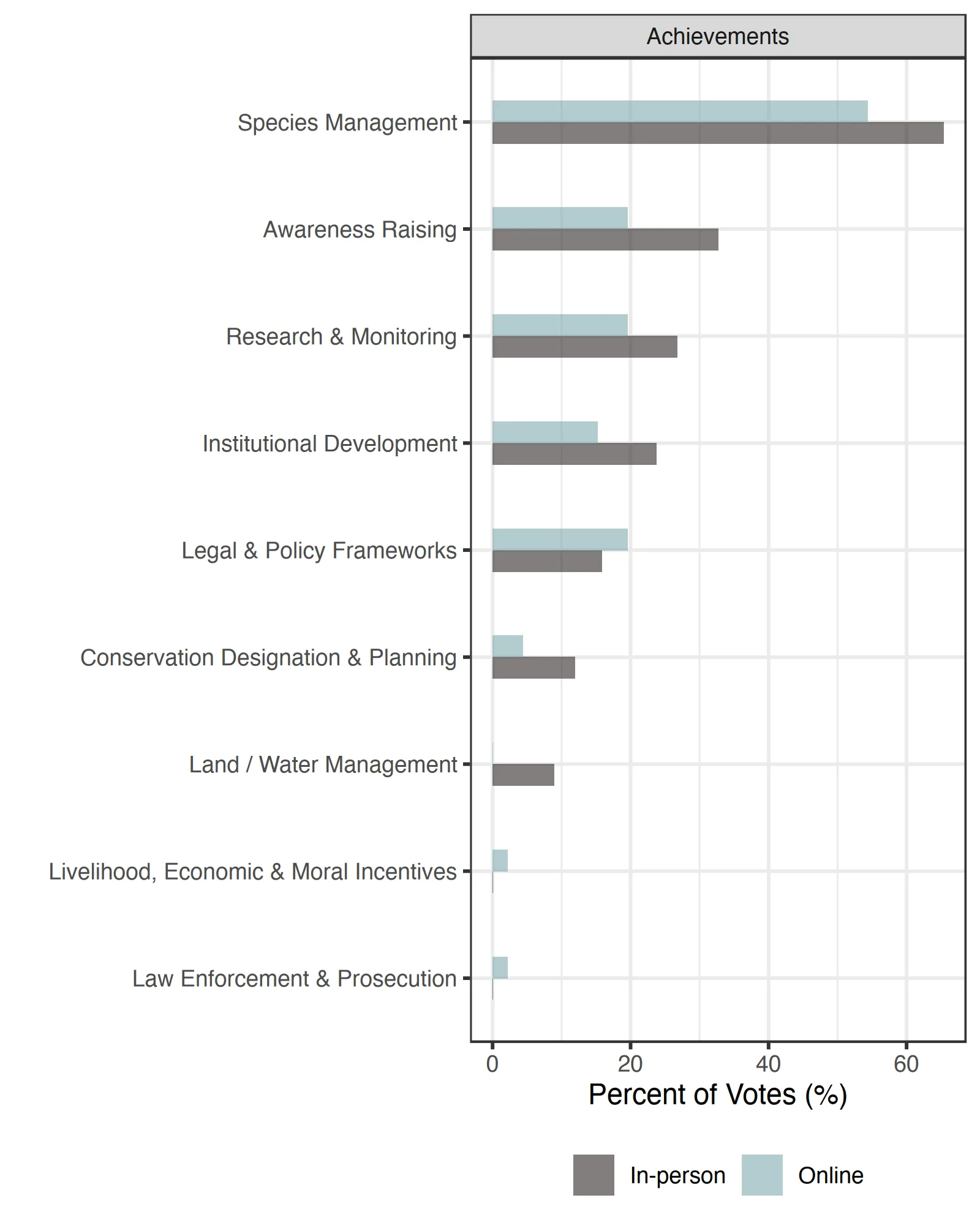
‘Species Management’ was identified as the top conservation/recovery achievement for Guloninae species over the past few years (Table 3, Figure 3). Specific examples from the ‘Species Re-Introduction & Translocation’ included: ‘In Scotland, over the last few decades, the pine marten (Martes martes) has recovered spectacularly, due to a reduction in persecution and a priori expansion in forest cover.’ and ‘Fishers are doing well following reintroduction several decades ago’. ‘Awareness Raising’ was another key achievement with specific examples of ‘Outreach & Communication’ including: ‘To show a different side of apex predators rewilding as potential control agents of mesocarnivores such as martens. In Mediterranean areas, the mesocarnivore most affected by the reintroduction of the Iberian lynx is the stone marten, which leads [them] to disappear from lynx territories. We have focused our informative tasks on informing people about the relevant role of mesocarnivores such as martens play in ecosystems and the importance of actions for mesopredator conservation and their ecological roles’. Achievements in ‘Research and Monitoring’ focused on new research efforts and findings, including ‘Annual monitoring of abundance’ while ‘Institutional Development’ achievements focused more on community involvement, new collaborations, and new funding, including: ‘We don’t have marten in the South East of England yet, but since we started our restoration project 2 years ago, we have successfully co-developed a strategy with a stakeholder working group made up of 30+ organisations and formed a very successful and collaborative project committee. Showcasing the importance of collaborating with others and including others in species reintroduction projects to reduce conflict, build trust and develop an evidence based project that benefits lots of wildlife and local people’ and ‘Funding secured for research’.
Threats
The largest threat to Guloninae around the globe was identified as ‘Climate Change’ through ‘Ecosystem Encroachment’ and ‘Changes in Temperature Regimes’ with specific examples including ‘Climate change’ and ‘Habitat loss via climate change (Table 4, Figure 4). Participants identified ‘Biological Resource Use’, predominantly through ‘Hunting & Collecting Terrestrial Animals’, with examples including ‘Persecution’, ‘Overharvesting / overhunting’, and ‘Poaching’.persecution, species overharvest, poaching), but also through ‘Logging & Wood Harvesting’ with examples including ‘Logging of taiga for sable’, ‘Large scale clear cutting’, and ‘No trees reach ages at which they develop cavities that fishers can use for natal dens’. Other threats identified at the jurisdictional scale included ‘Natural Systems Modification’, with specific examples including ‘Increased fire frequency and burn severity leading to reduction and total destruction of suitable habitat during these events. These fires move quickly and likely cause mortality of individuals within the fire footprint which could be detrimental to small and isolated populations’ and ‘Invasive & Problematic Species’, with specific examples including ‘In Hokkaido island, introduced Japanese marten, M. melampus, may compete with the sable, M. zibellina, for resources (habitat or food)’. Another key threat at the jurisdictional level was ‘Pollution’ with specific examples including ‘rodenticide’.
Actions
The management/conservation actions needed to further Guloninae conservation and recovery, and actions necessary to overcome the largest obstacles to Guloninae, in respondents’ jurisdictions, was clearly ‘Land/Water Management’ (Table 5, Figure 5), mostly referring to actions within the Specific examples of obstacles in the ‘Ecosystem & Natural Process (Re)Creation’ included: ‘Ultimately, there is no Martes conservation without proactive landscape level management’,
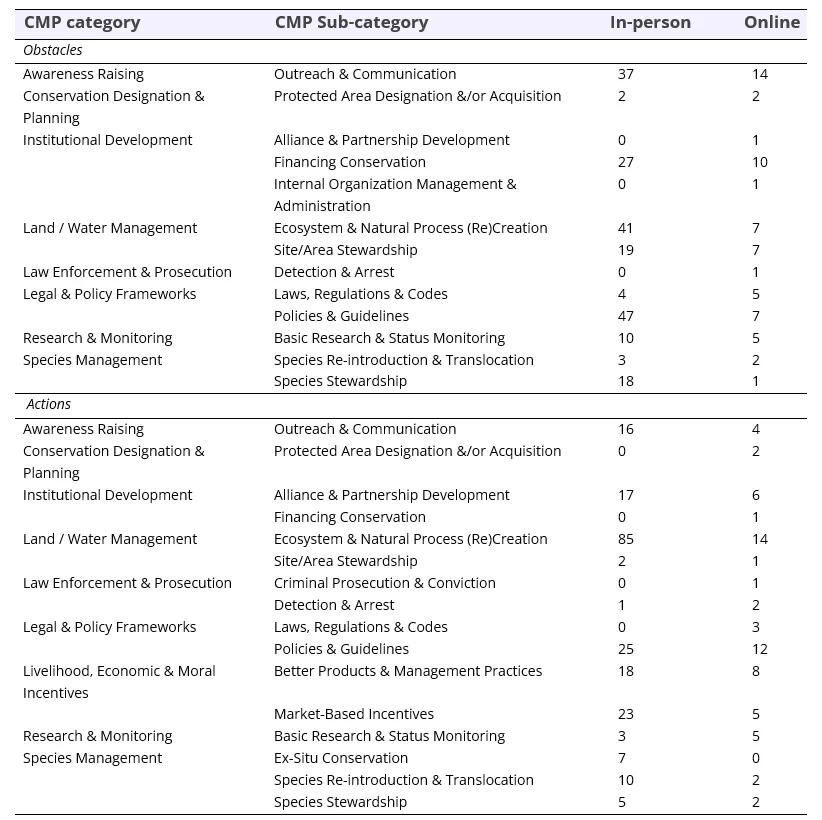
Table 5. Frequency of responses, by Conservation Measures Partnership (CMP) category and sub-category for in-person prioritization (n=101) and the online survey (n=46), answering the questions ‘What are the largest obstacles to Martes in your jurisdiction?’ and ‘What management/conservation actions are needed to further Martes conservation and recovery in your jurisdiction?
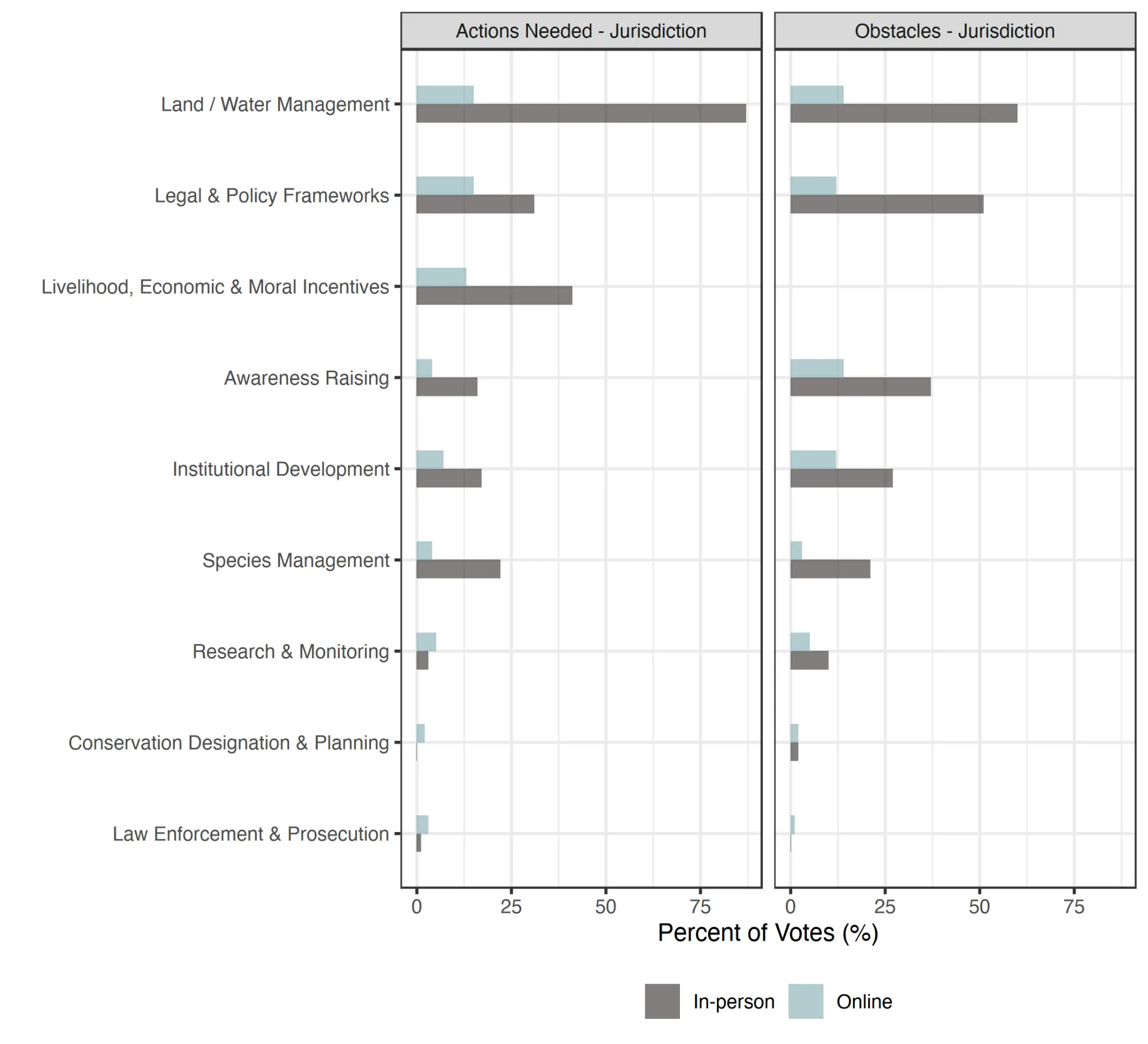
Figure 5. The management/conservation actions needed to further Guloninae conservation and recovery, and action necessary to overcome the largest obstacles to Guloninae, in respondents’ jurisdictions, as assessed by Martes Working Group members (online, n=46) and symposium attendees (in-person, n=101).
Information Needs
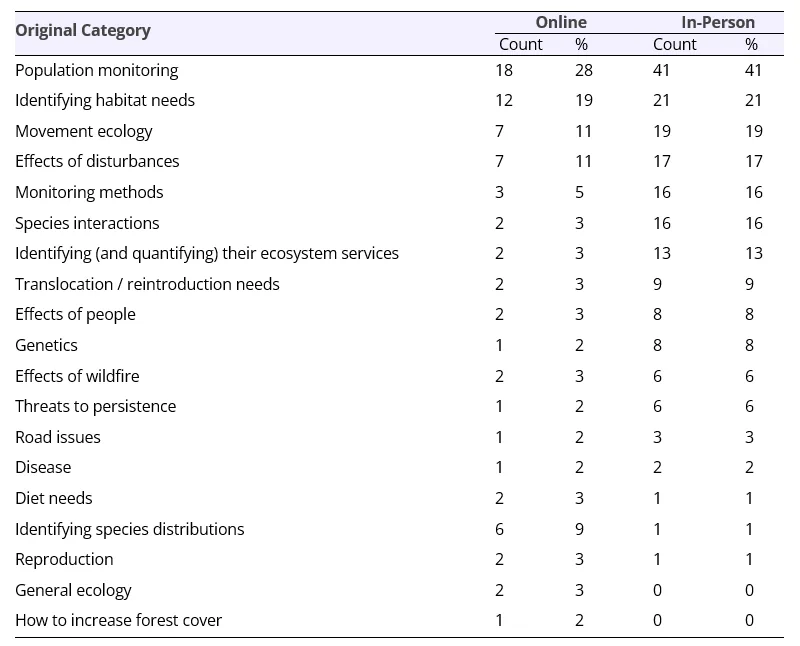
The largest information/research needed to further Guloninae conservation in participants’ jurisdictions was ‘Population monitoring’, with specific examples including ‘Baseline population estimates’, ‘Continuing monitoring for populations’ and ‘Accurate population estimates’ (Table 6). The second predominant information/research need was ‘Identifying habitat needs’, with specific examples including ‘Continue to gain knowledge on productivity in managed landscapes’ and ‘Thresholds for how little and the configuration of forest remaining for populations to survive’.
Overcoming Obstacles
The overarching action to overcome obstacles to Guloninae conservation that resonated most with participants was ‘Institutional Development’ (Figure 6) with a focus on ‘Alliance & Partnership Development’ (e.g., ‘Increasing dialogue between groups that support increasing species numbers and those who oppose it’ and ‘Data should be shared more readily, quickly and openly, so managers and practitioners can implement new science sooner’), ‘Financing Conservation’ (e.g., ‘More funding for research’), and ‘Internal Organization Management & Administration’ (e.g., ‘Recruit and invite a greater proportion of non-English speaking Martes researchers and offering more support if there are language barriers’). Other important actions were ‘Awareness Raising – Outreach and Communications’ (e.g., ‘Need to get better at refuting negative perceptions’ and ‘Promoting champions/ambassadors’); ‘Legal & Policy Frameworks – Policies and Guidelines’ (e.g., ‘Write policy statements/resolutions’, and ‘Work with decision makers’); and ‘Research & Monitoring – Basic Research & Status Monitoring’ (e.g., ‘Find better ways to identify limiting factors (e.g., how to find dens)’, and ‘How to continue, e.g., fur trapping, whilst continuing conservation’).
Replicate and scale wins
Similar to overcoming obstacles, the in-person discussion highlighted ‘Institutional Development’ (Figure 6) as the necessary actions for conservationists to replicate and scale wins, noting ‘Alliance & Partnership Development’ (e.g., ‘Identify issues practitioners are having and searching for a middle ground you can meet at’), ‘Financing Conservation’ (e.g., ‘Secure funding to support that information sharing’), and ‘Internal Organization Management & Administration’ (e.g., ‘MWG could provide step by step process of a successful process for activities (e.g., reintroductions)’). Other key actions identified were ‘Awareness Raising – Outreach & Communication’ (e.g., ‘Public knowledge and visibility’); ‘Research & Monitoring – Basic Research & Monitoring’ (e.g., ‘Piggybacking – get data from other species’ studies’); and ‘Species Management – Species Stewardship’ (e.g., ‘Local effort, replicated across multiple localities’).
Advance efforts
Similar to overcoming obstacles, the in-person discussion highlighted ‘Institutional Development’ (Figure 6) as the necessary actions for conservationists to replicate and scale wins, noting ‘Alliance & Partnership Development’ (e.g., ‘Identify issues practitioners are having and searching for a middle ground you can meet at’), ‘Financing Conservation’ (e.g., ‘Secure funding to support that information sharing’), and ‘Internal Organization Management & Administration’ (e.g., ‘MWG could provide step by step process of a successful process for activities (e.g., reintroductions)’). Other key actions identified were ‘Awareness Raising – Outreach & Communication’ (e.g., ‘Public knowledge and visibility’); ‘Research & Monitoring – Basic Research & Monitoring’ (e.g., ‘Piggybacking – get data from other species’ studies’); and ‘Species Management – Species Stewardship’ (e.g., ‘Local effort, replicated across multiple localities’).
Actions for near future success
Similar to overcoming obstacles, the in-person discussion highlighted ‘Institutional Development’ (Figure 6) as the necessary actions for conservationists to replicate and scale wins, noting ‘Alliance & Partnership Development’ (e.g., ‘Identify issues practitioners are having and searching for a middle ground you can meet at’), ‘Financing Conservation’ (e.g., ‘Secure funding to support that information sharing’), and ‘Internal Organization Management & Administration’ (e.g., ‘MWG could provide step by step process of a successful process for activities (e.g., reintroductions)’). Other key actions identified were ‘Awareness Raising – Outreach & Communication’ (e.g., ‘Public knowledge and visibility’); ‘Research & Monitoring – Basic Research & Monitoring’ (e.g., ‘Piggybacking – get data from other species’ studies’); and ‘Species Management – Species Stewardship’ (e.g., ‘Local effort, replicated across multiple localities’).
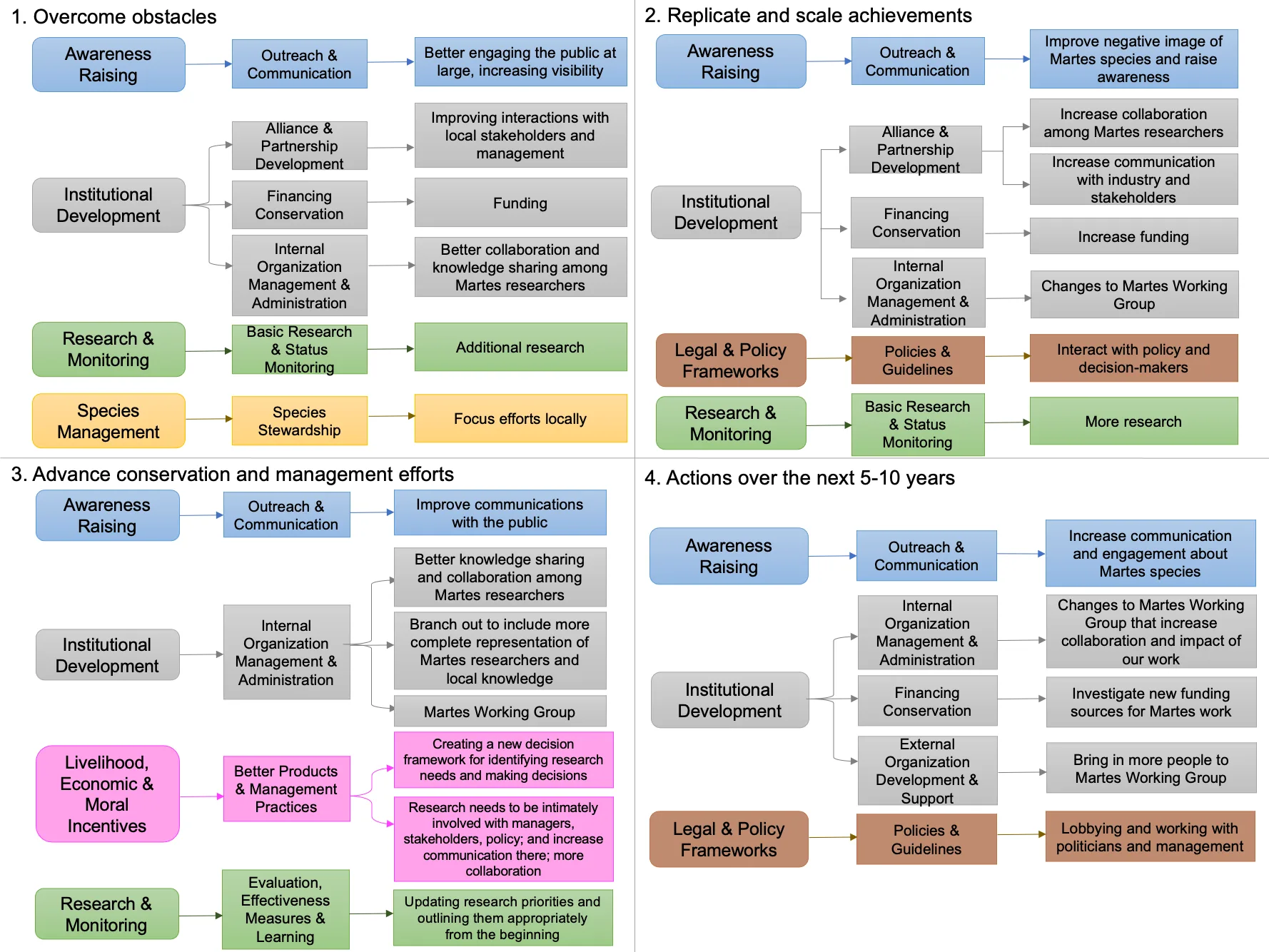
Figure 6. Common themes that emerged during the in-person discussion at the Martes Working Group symposium, in response to four questions (left to right, top to bottom): ’1. How can we overcome obstacles that exist for Martes conservation?’; ‘2. How can Martes conservationists replicate and scale wins?’; ‘3. How can research best advance conservation and management efforts?’; and ‘4. What actions can we take in the next year or two to lead to more success over the next 5-10 years?’.
Discussion
A key result from this research was the apparent unevenness of research efforts across Guloninae species. While some Guloninae species are well-researched, research efforts have been unevenly distributed among species (Proulx and Aubry 2020). For less studied Guloninae species there is a lack of natural history information that would benefit strategic and efficient conservation, such as information on habitat characteristics, amount of potential habitat within their range, home range size, and population dynamics. For species with a lack of contemporary knowledge, limited biologists, and for species that are locally concentrated (e.g., Nilgiri marten, yellow-throated marten, Japanese marten, and tayra), we were unable to summarize basic life history information, such as reproduction rates. Gathering experts for all species to describe our impression of the current status for these animals, even within an international symposium, was challenging. We will endeavor to build accessibility and outreach for tasks similar to our survey to Guloninae researchers outside of the core of individuals within North America and Europe.
A strength of our research was in finding several common themes amongst participants’ perspectives on Guloninae conservation and relative agreement in primary actions desired for conservation and recovery measures. This convergence bodes well for our ability to effectively identify common threats to, and pathways towards, conservation of Guloninae species. While each jurisdiction and species will necessitate a local understanding of threats and actions for recovery, researchers worldwide can base research and conservation action on global lessons learned. For example, fisher researchers from Canada can look to the United Kingdom and identify how strategies for raising awareness of the European pine marten evolved into successful species stewardship (e.g., MacPherson et al. 2024). The groups targeted in Canada will differ from those targeted in the UK, but general strategies on raising awareness can be shared.
The specific primary threats to the conservation of Guloninae species varies across different parts of their range, necessitating custom approaches to conservation strategies. For example, the yellow-throated marten is threatened by habitat fragmentation and road traffic in South Korea (Kang et al. 2023), incidental catch during the hunting of other game species in Russia (Oleynikov et al. 2022; Yudin and Yudina 2022), and poaching in Nepal and India (Basnet and Rai 2020; Malick 2015). A deep understanding of the local threats to species’ persistence should always be considered when determining recovery strategies, which would be facilitated by increasing the number of regional level Red List assessments.
A limitation to this research was that survey responses, prioritization and in-person discussions were not species-specific. While we asked respondents and symposium attendees to consider their study species we did not ask for specifications when responding, prioritizing, or discussing. As many respondents and attendees studied multiple species it was not possible to attribute responses to specific species, unless clearly noted in the response (i.e., not just a reference to ‘marten’). In future studies we recommend linking responses to species to allow specification and aggregation. We incorporate examples provided in the results with the broader literature for a more comprehensive discussion. We recommend that a future publication(s) provides a more in-depth review of Guloninae species, where more specific threats, management concerns, and proposed actions are laid out for each species.
Conservation achievement: Species management
By far, the biggest recent “achievement” for Guloninae species conservation was identified as species management, particularly species stewardship, reintroduction, and translocation (Powell et al. 2012). As noted by respondents, Newfoundland marten populations are recovering (Hearn and Durocher 2023) and European pine martens are expanding their range in Scotland (Sainsbury et al. 2019) and south of West Siberia (Herrero et al. 2016; Monakhov 2022) Pine martens have been successfully translocated in Wales and England (MacPherson and Wright 2021). Fishers appear to be doing well in the Eastern US, following reintroduction several decades ago and also were translocated to the Olympic Mountains and Cascade Range in Washington State, USA (Happe et al. 2020; Lewis et al. 2022) and to northern Sierra Nevada in California, USA (Green et al. 2022). The timing and approach for such translocations or augmentations need to be considered (e.g., Facka et al. 2016).
A notable conservation achievement that was minimally expressed by our participants was the use of protected area designations or other conservation design and planning actions. Protected natural areas continue to play an important role for Guloninae species (McDonald and Boucher 2011; Proulx and Aubry 2020), especially for those sensitive to human intervention and infrastructure (May 2007; Fisher et al. 2022). National parks and large natural reserves appear to be most beneficial when they encompass suitable areas and are protected with adequate law enforcement. These areas will be strategic for upcoming Guloninae management plans (Proulx and Aubry 2020). For example, successful sable restoration in Russia was closely linked to the establishment of protected areas (Chernikin 2006). Mass reintroductions in the mid-20th century ensured the creation of several large populations of sable in Western and Yenisei Siberia, the Republic of Yakutia-Sakha, and other areas in the east of the range (Powell et al. 2012; Monakhov 2015). Perhaps these conservation actions were not considered achievements because respondents did not consider them as ‘recent’, but long-term planning will continue to be a cornerstone of conservation for animals requiring large spatial areas.
Conservation achievement: Awareness raising
Another conservation achievement was awareness raising, including overcoming negative perceptions of these species through outreach and communications. Extensive community engagement programs were built into the British reintroductions of European pine marten to England and Wales (Litchfield 2019). These programs aimed to educate and raise awareness of the species, and mitigate potential conflicts within the local community. The reintroduction projects received high levels of support, and the community greatly valued these engagement opportunities (MacPherson et al. 2014; Litchfield 2019). As populations expand to new areas, additional projects are in place to continue raising awareness amongst new communities, and build on successes (e.g., ‘Martens on the Move’ campaign by the Vincent Wildlife Trust). Communicating the importance of mesopredator conservation and their ecological roles is another key action, especially where the reintroduction of endangered predators, such as the Iberian lynx (Lynx pardinus), can lead to exclusion of stone marten (e.g., Burgos et al. 2023). While raising awareness was a conservation achievement, the lack of awareness was identified as an obstacle to Guloninae species conservation.
Identified threat: Climate change
Symposium attendees agreed that the biggest global threat to Guloninae species will be climate change, including changes in temperature, reductions in snowpack, and the large-scale effects of ecosystems shifting and impinging on other species and ecosystems. Ten of the eleven Guloninae species were found to be either moderately or highly vulnerable to the climate and ecological emergency, particularly the impacts of increasing temperatures and wildfires (Birks et al. 2025). The majority of Guloninae species occur at northern latitudes and in temperate regions—areas that are predicted to experience the greatest increases in temperature and dramatic shifts in vegetation communities—so they may be at greater risk and experience adverse effects. The effects of climate change on the distribution of Guloninae species have been observed. Examples that appear concerning for specific Guloninae species include wildfire and drought impacting fisher and marten populations (Green et al. 2022; Kordosky et al. 2021), changing snow levels influencing wolverine, American marten, and Pacific marten (McKelvey et al. 2011; Suffice et al. 2017; Aubry et al. 2023; Barsugli et al. 2020; Moriarty et al. 2015; Moriarty et al. 2019; Martin et al. 2020; Colella et al. 2024) and elevated temperatures that may negatively influence beech marten, European pine marten, and yellow-throated marten (Dutta et al. 2022; Wereszczuk et al. 2021; Wereszczuk and Zalewski 2023).
Identified threat: Biological resource use
Results indicated that the predominant threat across populations was biological resource use, with nearly equal impacts from hunting/trapping and habitat changes from timber harvesting. The IUCN identifies hunting/trapping as one of the population threats for each of the listed Guloninae species, with the exception of the Japanese marten and yellow-throated marten. Fur harvesting can be challenging to sustain due to the relatively low reproductive rates and large home ranges of Guloninae species (Powell and Thompson 1993; Aubry and Proulx 2017; Barrueto et al. 2022; Mowat et al. 2020). Direct persecution can lead to a decline in their abundance (e.g., Fogarty et al. 2022), as evidenced by the recent history of many of these species (Sainsbury et al. 2019; Bakeyev and Sinitsyn 1994; Monakhov 2011). The increase in the threat of overharvesting is directly linked to periods of growth in their economic value and human encroachment.
Forest harvesting and habitat loss continues to be a concern for Guloninae species, but is particularly important to the one Guloninae species listed as Vulnerable, i.e., the Nilgiri marten in western India (Shameer et al. 2023). The impact of forest harvesting and habitat management on populations can be difficult for jurisdictions to manage within the borders of one or a few countries, but it may be easier than for species inhabiting many countries, where coordinating and implementing unified conservation programs is much more challenging (Mason et al. 2020).
Identified action: Land/water management
Participants focused primarily on actions that targeted restoration or reduced stress (i.e., land/ water management) with specific focus on restoring missing or severely degraded ecosystems and mitigating stresses for sites and/or ecosystem targets. Woodland creation and forest restoration were consistently mentioned as the most pressing action needed to recover Guloninae species at the jurisdictional level. Many Guloninae species are forest dependent and features from forest cover (e.g., rest and den sites, available prey) are integral to their survival and reproduction (e.g., Bianchi et al. 2021; Kordosky et al. 2021; Balestrieri et al. 2021). For example, in the Central Interior of British Columbia, even a small increase in forest harvesting area decreases the likelihood of a resident fisher including that area as part of their home range, which has dramatic implications for landscapes inundated with timber harvesting to support sustainable fisher populations (Weir and Corbould 2010). It is not surprising that the key action that emerged for Guloninae species recovery was to recover and restore habitat, considering that the key jurisdictional threat included logging and wood harvesting.
Information needs: Population monitoring
Understanding the status and trends of Guloninae species is critical for charting recovery actions, and our research indicates that population monitoring is an important information need for many Guloninae species. For example, Pacific martens in Washington State, USA, occur as two distinct currently unconnected populations, one in the Cascade Range and the other in the Coastal Mountains of the Olympic Peninsula. Recent genetic analysis indicates that there historically was greater connectivity between these two populations (Schwartz et al. 2020), but that Pacific martens have experienced a decline on the Olympic Peninsula of Washington (Zielinski et al. 2001; Moriarty et al. 2019). Their rarity and the challenges inherent in monitoring them have indicated a limited distribution and potentially critically low numbers for Pacific ‘coastal’ marten throughout the Olympic Peninsula (Moriarty et al. 2019; Howell et al. 2020). Rapid changes to habitat, e.g., massive wildfires in different parts of the world, also necessitate the continuing need for monitoring. Some species may adapt to the changing conditions or but areas that increase in temperature, decrease in forest cover, and experience high severity fire may not be suitable for Guloninae populations (e.g., Collier 2024).
In some instances, the continued commercial harvest of mammals hinges on the demonstration of stable population status and numbers. For example, in Russia sable exploitation is managed through annual hunting limits, serving as a form of resource protection. Population stability is monitored through annual winter route surveys conducted by organizations managing hunting grounds under regional hunting departments’ control. Data from these surveys inform decisions on hunting exploitation and conservation measures. For other Guloninae species, however, quotas or hunting/trapping limits are not imposed as they are not deemed to be of concern to resource conservation. Nonetheless, banning hunting of yellow-throated martens has been suggested in certain areas of the Far East (Oleynikov and Tkachenko 2013; Oleynikov et al. 2022; Yudin 2022). In Sweden and Norway, European pine martens are subjected to predator control measures in some areas to enhance forest grouse populations, and there is a need for proper management and monitoring to avoid local decimation where this occurs (Helldin 2000). Novel attempts to reduce conflicts are being executed (e.g., Bamber et al. 2024).
Conclusions
Our approach of collecting expert opinion to identify threats to Guloninae species was successful in highlighting unifying themes across species, particularly climate change and biological resource use (hunting/trapping and forest harvesting). The actions needed to overcome obstacles and further conservation/recovery were more nuanced, but respondents identified unifying themes of improving land management and increasing species protection. With all but one Guloninae species listed as Least Concern by the IUCN, elevating the importance of conservation actions with decision-makers can be difficult and providing public outreach regarding a species’ vulnerability is challenging. A common and recurring theme during the in-person discussions was the need for actions that create the conditions necessary for conservation efforts to succeed, in particular the actions that enable institutions to support conservation work (e.g., by increasing funding, collaboration, and communication).
Having a plan for the conservation and recovery of Guloninae species is vital for gathering support for species recovery among lawmakers, land managers, and other decision-makers, prioritizing conservation actions, obtaining necessary financial and social support for species recovery, connecting networks of partners to more efficiently accomplish goals and objectives, and maintaining and connecting habitats. This collaboration intends to create an international Guloninae Action Plan (GAP) that is updated every five years to keep the needs of Guloninae species current, and allow progress to be reported and re-prioritization of actions to occur. Lessons learned from this research will inform the GAP; i.e., the collaboration will extend to less well studied Guloninae species and include researchers outside of North America and Europe. It will highlight actions that benefit specific species and actions that benefit multiple Guloninae species. The GAP will be introduced at the 2026 Martes Working Group Symposium, and will be accessible via the Martes Working Group website (https://www.martesworkinggroup.org/). Without effective conservation planning, investments in conservation for these vital species may continue to suffer from ineffective strategies, poor return on conservation investments, and potential loss of these species from ecosystems. The outcome of this assessment will be an evaluation of common threats, successful and unsuccessful conservation and recovery approaches, and guidance to help researchers, conservation program managers, and regulatory agencies achieve effective conservation of Guloninae. We hope our combined efforts, along with willing land managers, can support these species. We would like members of Guloninae to continue to provide their ecosystem roles. Our goal to efficiently guide future research and provide support towards conservation efforts aims to maximize the persistence of Guloninae species.
Acknowledgments
This research would not have been possible without the support of the Martes Working Group, particularly the organizers and attendees of the 2023 symposium. We thank Alli Fitzmorris for providing the Guloninae sketches and Jeff Dixon for providing the graphical abstract.
During the writing of this manuscript we lost a vital and well-respected member of the Martes community: Vladimir Genrikhovich Monakhov tragically passed away on June 7, 2024. We are forever grateful for his research contributions and wisdom on sable and other Guloninae species.
Author Contributions
Conceptualization: All
Data curation: JMB, JM, SY
Formal analysis: JMB
Investigation: SY
Methodology: JMB, SY
Visualization: ERS, AYO, JMB, PH
Writing – original draft: All
Writing – review & editing: All
Data Availability
Full data and results can be found in the datasets below.
- Raw data from online survey
- Top themes from in-person voting for discussion
- Raw data from in-person discussion
- Collated data from in-person voting and discussion following IUCN Unified Classifications of Direct Threats and Conservation Actions
Transparent Peer Review
Results from the Transparent Peer Review can be found here.
Recommended Citation
Burgar, J. M., Yaeger, S., Anderson, E., Brainerd, S., Chabaud, N., Cotey, S. R., Croose, E., Fisher, J. T., Hansen, I. J., Hapeman, P., Hofmeester, T. R., Howell, B. L., Kingscott, J., MacPherson, J., Manzo, E., Monakhov, V. G., Poirson, C., Oleynikov, A. Y., Scopes, E. R., Smith, H., & Spencer, W. D. (2025). A path towards the conservation and recovery of Guloninae species worldwide. Stacks Journal: 25007. https://doi.org/10.60102/stacks-25007
References
Abramov, A. V., A. Kranz, J. Herrero, A. Choudhury, and T. Maran. 2016. “The IUCN Red List of Threatened Species 2016: e.T29672A45202514.” IUCN. https://doi.org/10.2305/iucn.uk.2016-1.rlts.t29672a45202514.en.
Abramov, A.V., Kaneko, Y. & Masuda, R. 2015. Martes melampus. The IUCN Red List of Threatened Species 2015: e.T41650A45213228. https://dx.doi.org/10.2305/IUCN.UK.2015-4.RLTS.T41650A45213228.en
Ashbrook, Sarah, and Paul Hapeman. 2024. “American Marten Occupancy and Activity Patterns at the Southern Extent of Their Range in the Eastern United States.” Ecology and Evolution 14 (2): e10904.
Aubry, Keith B., Catherine M. Raley, Andrew J. Shirk, Kevin S. McKelvey, and Jeffrey P. Copeland. 2023. “Climatic Conditions Limit Wolverine Distribution in the Cascade Range of Southwestern North America.” Canadian Journal of Zoology 101 (2): 95–113.
Aubry, Keith B., and Gilbert Proulx. 2017. “The Martes Complex: A Monophyletic Clade That Shares Many Life-History Traits and Conservation Challenges.” In The Martes Complex in the 21st Century: Ecology and Conservation., edited by Andrzej Zalewski, Izabela Wierzbowska, Keith B. Aubry, Johnny D. S. Birks, Declan T. O’Mahony, and Gilbert Proulx, 3–24. Białowieża, Poland: Mammal Research Institute, Polish Academy of Sciences.
Badmaev, A. A. 2003. “Sable in the Traditional Culture of the Buryats.” Bulletin of Archaeology, Anthropology and Ethnography 2 (61): 151–56.
Bakeyev, N. N., and A. A. Sinitsyn. 1994. “Status and Conservation of Sables in the Commonwealth of Independent States.” In Martens, Sables, and Fishers: Biology and Conservation., edited by S. W. Buskirk, A. S. Harestad, M. G. Raphael, and R. A. Powell, 246–54. Ithaca, New York: Cornell University Press.
Balestrieri, A., A. Mosini, F. Fonda, M. Piana, P. Tirozzi, A. Ruiz-González, E. Capelli, et al. 2021. “Spatial Ecology of the Stone Marten in an Alpine Area: Combining Camera-Trapping and Genetic Surveys.” Mammal Research 66 (2): 267–79.
Balestrieri, A., Remontia, L., Ruiz-González, A., Gómez-Moliner, B.J., Vergara, M. and C. Prigoni, Range expansion of the pine marten (Martes martes) in an agricultural landscape matrix (NW Italy). Mamm. Biol. (2009), doi:10.1016/j.mambio.2009.05.003
Ballejo, Fernando, Pablo Plaza, Agustina di Virgilio, Mauro Lucherini, Pablo Gáspero, María M. Guerisoli, Andrés Novaro, Martín Funes, and Sergio A. Lambertucci. 2022. “Desentrañando Las Interacciones Negativas Entre Humanos, Mamíferos Carnívoros Y Rapaces En América Del Sur.” Ecologia Austral 32 (2): 620–37.
Bamber, Jack A., Kenny Kortland, Chris Sutherland, Ana Payo-Payo, and Xavier Lambin. 2024. “Evaluating Diversionary Feeding as a Method to Resolve Conservation Conflicts in a Recovering Ecosystem.” The Journal of Applied Ecology 61 (8): 1968–78.
Barrueto, Mirjam, Anne Forshner, Jesse Whittington, Anthony P. Clevenger, and Marco Musiani. 2022. “Protection Status, Human Disturbance, Snow Cover and Trapping Drive Density of a Declining Wolverine Population in the Canadian Rocky Mountains.” Scientific Reports 12 (1): 17412.
Barsugli, Joseph J., Andrea J. Ray, Ben Livneh, Candida F. Dewes, Aaron Heldmyer, Imtiaz Rangwala, John M. Guinotte, and Stephen Torbit. 2020. “Projections of Mountain Snowpack Loss for Wolverine Denning Elevations in the Rocky Mountains.” Earth’s Future 8 (10): e2020EF001537. https://doi.org/10.1029/2020ef001537.
Basnet, Hari, and Anu Rai. 2020. “An Update on the Distribution of Yellow-Throated Marten Martes flavigula in Nepal.” Nepalese Journal of Zoology 4 (2): 147–51.
Bianchi, Rita, Julianna M. A. Jenkins, Damon B. Lesmeister, Jéssica Abonízio Gouvea, Clarice Silva Cesário, Larissa Fornitano, Mateus Yan de Oliveira, Kimberly Danielle Rodrigues de Morais, Renan Lieto Alves Ribeiro, and Matthew E. Gompper. 2021. “Tayra (Eira barbara) Landscape Use as a Function of Cover Types, Forest Protection, and the Presence of Puma and Free‐ranging Dogs.” Biotropica 53 (6): 1569–81.
Bilandžić, Nina, Danko Dežđek, Marija Sedak, Maja Dokić, Božica Solomun, Ivana Varenina, Zorka Knežević, and Alen Slavica. 2010. “Concentrations of Trace Elements in Tissues of Red Fox (Vulpes Vulpes) and Stone Marten (Martes foina) from Suburban and Rural Areas in Croatia.” Bulletin of Environmental Contamination and Toxicology 85 (5): 486–91.
Birks, J., Johnstone, S., Snell, E., & MacPherson, J. (2025). What is the future for The Martes Complex (Guloninae) in the face of climate change and ecological breakdown? Stacks Journal: 25001. https://doi.org/10.60102/stacks-25006.
Bischof, Richard, Cyril Milleret, Pierre Dupont, Joseph Chipperfield, Mahdieh Tourani, Andrés Ordiz, Perry de Valpine, et al. 2020. “Estimating and Forecasting Spatial Population Dynamics of Apex Predators Using Transnational Genetic Monitoring.” Proceedings of the National Academy of Sciences of the United States of America 117 (48): 30531–38.
Bonaccorso, Frank J., William E. Glanz, and Clark M. Sandford. 1980. “Feeding Assemblages of Mammals at Fruiting Dipteryx Panamensis (Papilionaceae) Trees in Panama: Seed Predation, Dispersal, and Parasitism.” Revista de Biologia Tropical 28 (1): 61–72.
Brainerd, Scott M., and Jørund Rolstad. 2002. “Habitat Selection by Eurasian Pine Martens Martes martes in Managed Forests of Southern Boreal Scandinavia.” Wildlife Biology 8 (4): 289–97.
Burgos, Tamara, Javier Salesa, Jose María Fedriani, Gema Escribano-Ávila, José Jiménez, Miha Krofel, Inmaculada Cancio, Javier Hernández-Hernández, Javier Rodríguez-Siles, and Emilio Virgós. 2023. “Top-down and Bottom-up Effects Modulate Species Co-Existence in a Context of Top Predator Restoration.” Scientific Reports 13 (1): 4170.
Buskirk, S. W., and R. A. Powell. 1994. “Habitat Ecology of Fishers and American Martens.” In Martens, Sables, and Fishers: Biology and Conservation., edited by S. W. Buskirk, A. S. Harestad, and M. G. Raphael, 283–96. Ithaca, NY: Cornell University Press
Camargo-Sanabria, Angela A., and Eduardo Mendoza. 2016. “Interactions between Terrestrial Mammals and the Fruits of Two Neotropical Rainforest Tree Species.” Acta Oecologica 73 (May): 45–52.
Chavez, C. 2014. “Tayra (Eira barbara).” In Mammals of Mexico, edited by G. Ceballos. Baltimore,MD.: Johns Hopkins University Press.
Chernikin, E. M. 2006. “Ecology of the Sable (Martes zibellina Lunneus, 1758) in the Barguzin Nature Reserve.” Ulan-Ude: Buryat State University Publishing.
Cheveau, Marianne, Louis Imbeau, Pierre Drapeau, and Louis Belanger. 2013. “Marten Space Use and Habitat Selection in Managed Coniferous Boreal Forests of Eastern Canada.” The Journal of Wildlife Management 77 (4): 749–60.
Chiang, Po-Jen, K. Pei, M. Vaughan, and Ching‐feng Li. 2012. “Niche Relationships of Carnivores in a Subtropical Primary Forest in Southern Taiwan.” Zoological Studies 51 (4): 500–511.
Chutipong, W., J. W. Duckworth, Timmins R. Choudhury, A Abramov A, S. Roberton, B. Long, H. Rahman, A. Hearn, V. Dinets, and D. H. A. Willcox. 2016. “Martes flavigula. The IUCN Red List of Threatened Species 2016: e.T41649A45212973.” https://dx.doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41649A45212973.en
Colella, Jocelyn P., Nicholas A. Freymueller, Danielle M. Land, Ben J. Wiens, Karen D. Stone, and Joseph A. Cook. 2024. “Ecological Displacement in a Rocky Mountain Hybrid Zone Informs Management of North American Martens (Martes).” Landscape Ecology 39 (7), 125.
https://doi.org/10.1007/s10980-024-01915-y.
Collier, C. J. 2024. “Fire Severity Mediates Marten and Fisher Occurrence: Impacts of the Dixie Fire on a Carnivore Community.” Masters, California State Polytechnic University, Humboldt.
Copeland, J. P., K. S. McKelvey, K. B. Aubry, A. Landa, J. Persson, R. M. Inman, J. Krebs, et al. 2010. “The Bioclimatic Envelope of the Wolverine (Gulo gulo): Do Climatic Constraints Limit Its Geographic Distribution?” Canadian Journal of Zoology 88 (3): 233–46.
Copeland, J. P., and T. E. Kucera. 1997. “Wolverine (Gulo gulo).” In Mesocarnivores of Northern California: Biology, Management, and Survey Techniques, Workshop Manual. August 12-15, 1997, Humboldt State Univ., Arcata, CA., edited by John E. Harris and Chester V. Ogan., 23–33. Arcata, CA: The Wildlife Society, California North Coast Chapter.
Cui, Jifa, Yaqian Zhang, Jinyu Guo, Nan Wu, and Youbing Zhou. 2023. “Conflicting Selection Pressures on Seed Size and Germination Caused by Carnivorous Seed Dispersers.” Integrative Zoology 18 (5): 799–816.
Danneyrolles, Victor, Arseneault, D., and Y. Bergeron. 2016. “Long-Term Compositional Changes Following Partial Disturbance Revealed by the Resurvey of Logging Concession Limits in the Northern Temperate Forest of Eastern Canada.” Canadian Journal of Forest Research. Journal Canadien de La Recherche Forestiere 46 (May): 943–49.
Dawson, Natalie G., Colella, Jocelyn P., Small, Maureen P., Stone, Karen D., Talbot, Sandra L. , and Joseph A. Cook. 2017. Historical biogeography sets the foundation for contemporary conservation of martens (genus Martes) in northwestern North America, Journal of Mammalogy, Volume 98, Issue 3: 15–730. https://doi.org/10.1093/jmammal/gyx047
Dawson, Natalie G., and Joseph A. Cook. 2012. “Behind the Genes: Diversification of North American Martens (Martes americana and M. caurina).” In Biology and Conservation of Martens, Sables, and Fishers, edited by K. B. Aubry, W. J. Zielinski, M. G. Raphael, G. Proulx, and S. W. Buskirk, 23–38. Ithaca, NY: Cornell University Press.
Do Linh San, Emmanuel, Jun J. Sato, Jerrold L. Belant, and Michael J. Somers. 2022. “The World’s Small Carnivores.” In Small Carnivores, 1–38.
Douglas, C. W. and Strickland, M. A. 1987. “Fisher.” In Wild Furbearer Management and Conservation in North America, edited by M. Novak, 510–29. Ontario Ministry of Natural Resources.
Dutta, Ritam, Tanoy Mukherjee, Amira Sharief, Hemant Singh, Vineet Kumar, Bheem Dutt Joshi, Dhriti Banerjee, Mukesh Thakur, and Lalit Kumar Sharma. 2022. “Climate Change May Plunder the Facultative Top Predator Yellow-Throated Marten from the Hindu-Kush Himalayan Region.” Ecological Informatics 69 (July): 101622.
Elmeros, Morten, Pia Lassen, Rossana Bossi, and Christopher J. Topping. 2018. “Exposure of Stone Marten (Martes foina) and Polecat (Mustela putorius) to Anticoagulant Rodenticides: Effects of Regulatory Restrictions of Rodenticide Use.” The Science of the Total Environment 612 (January): 1358–64.
Facka, Aaron N., Jeffrey C. Lewis, Patricia Happe, Kurt Jenkins, Richard Callas, and Roger A. Powell. 2016. “Timing of Translocation Influences Birth Rate and Population Dynamics in a Forest Carnivore.” Ecosphere 7 (1): e01223.
Facka, Aaron N., and Roger A. Powell. 2021. “Intraspecific Competition, Habitat Quality, Niche Partitioning, and Causes of Intrasexual Territoriality for a Reintroduced Carnivoran.” Frontiers in Ecology and Evolution 9 (November). https://doi.org/10.3389/fevo.2021.734155.
Fernandez, A.L.R. 2024. “mdd: Download mammal shapefiles of the world.” R package version 0.1.0, https://github.com/alrobles/mdd.
Fisher, J. T., B. Anholt, S. Bradbury, and M. Wheatley. 2013. “Spatial Segregation of Sympatric Marten and Fishers: The Influence of Landscapes and Species‐scapes.” Ecography 36(2): 240-248. https://onlinelibrary.wiley.com/doi/pdf/10.1111/j.1600-0587.2012.07556.x.
Fisher, Jason T., Sean Murray, Mirjam Barrueto, Kathleen Carroll, Anthony P. Clevenger, Doris Hausleitner, William Harrower, et al. 2022. “Wolverines (Gulo gulo) in a Changing Landscape and Warming Climate: A Decadal Synthesis of Global Conservation Ecology Research.” Global Ecology and Conservation 34 (April): e02019.
Fisher, Jason T., and A. Cole Burton. 2018. “Wildlife Winners and Losers in an Oil Sands Landscape.” Frontiers in Ecology and the Environment 16 (6): 323–28.
Fogarty, R. D., R. D. Weir, E. C. Lofroth, and K. W. Larsen. 2022. “Trapping Mortality Accelerates the Decline of the Fisher, an Endangered Mesocarnivore, in British Columbia, Canada.” Endangered Species Research 49 (September): 1–12.
Fogarty, Rory D., Richard D. Weir, and Karl W. Larsen. 2025. “Forestry, Fire, and Fur: Factors Influencing Densities of Endangered Fishers (Pekania pennanti) in Central British Columbia, Canada.” The Journal of Wildlife Management 89 (4). https://doi.org/10.1002/jwmg.70010.
Gabriel, Mourad W., Leslie W. Woods, Greta M. Wengert, Nicole Stephenson, J. Mark Higley, Craig Thompson, Sean M. Matthews, et al. 2015. “Patterns of Natural and Human-Caused Mortality Factors of a Rare Forest Carnivore, the Fisher (Pekania pennanti) in California.” PloS One 10 (11): e0140640.
Galef, Bennett G., Russell A. Mittermeier, and Robert C. Bailey. 1976. “Predation by the Tayra (Eira barbara).” Journal of Mammalogy 57 (4): 760–61.
Grattarola, Florencia, Kateřina Tschernosterová, and Petr Keil. 2024. “A Continental-Wide Decline of Occupancy and Diversity in Five Neotropical Carnivores.” Global Ecology and Conservation 55 (e03226): e03226.
Green, David S., Aaron N. Facka, Kevin P. Smith, Sean M. Matthews, and Roger A. Powell. 2022. “Evaluating the Efficacy of Reintroducing Fishers (Pekania pennanti) to a Landscape Managed for Timber Production.” Forest Ecology and Management 511 (May): 120089.
Green, David S., Marie E. Martin, Roger A. Powell, Eric L. McGregor, Mourad W. Gabriel, Kristine L. Pilgrim, Michael K. Schwartz, and Sean M. Matthews. 2022. “Mixed‐severity Wildfire and Salvage Logging Affect the Populations of a Forest‐dependent Carnivoran and a Competitor.” Ecosphere 13 (1): e03877. https://doi.org/10.1002/ecs2.3877.
Grotta-Neto, Francisco, Michel C. H. Mello, Raphael C. Mello, Itiberê P. Bernardi, Eduardo Carrano, and Fernando C. Passos. 2021. “The Role of Tayra (Eira barbara) as Predator of Medium and Large‐sized Mammals.” Austral Ecology 46 (2): 329–33.
Gura, A. V. 1997. “Animal Symbolism in the Slavic Folk Tradition. Moscow: Indrik.” Ecography 22: 324–36.
Gustine, David D., Katherine L. Parker, Roberta J. Lay, Michael P. Gillingham, and Douglas C. Heard. 2006. “Calf Survival of Woodland Caribou in a Multi-Predator Ecosystem.” Wildlife Monographs 165 (December): 1–32.
Hapeman, Paul, Emily K. Latch, Jennifer A. Fike, Olin E. Rhodes, and C. William Kilpatrick. 2011. “Landscape Genetics of Fishers (Martes pennanti) in the Northeast: Dispersal Barriers and Historical Influences.” The Journal of Heredity 102 (3): 251–59.
Hapeman, Paul, Emily K. Latch, Olin E. Rhodes, Brad Swanson, and C. William Kilpatrick. 2017. “Genetic Population Structure of Fishers (Pekania pennanti) in the Great Lakes Region: Remnants and Reintroductions.” Canadian Journal of Zoology 95 (11): 869–76.
Happe, Patricia J., Kurt J. Jenkins, Rebecca M. Mccaffery, Jeffrey C. Lewis, Kristine L. Pilgrim, and Michael K. Schwartz. 2020. “Occupancy Patterns in a Reintroduced Fisher Population during Reestablishment.” The Journal of Wildlife Management 84 (2): 344–58.
Hearn, B. J., and A. Durocher. 2023. “Habitat Availability and Population Size for American Marten (Martes americana atrata) on the Island of Newfoundland. Internal Research and Monitoring Report WLRM-2023-01.” Wildlife Division, Department of Fisheries, Forestry and Agriculture, Government of Newfoundland and Labrador.
Heim, N., Fisher, J. T., Clevenger, A., Paczkowski, J. and J. Volpe. 2017. “Cumulative Effects of Climate and Landscape Change Drive Spatial Distribution of Rocky Mountain Wolverine (Gulo gulo L.).” Ecology and Evolution 7 (21): 8903–14.
Helldin, J. O. 1999. “Diet, Body Condition, and Reproduction of Eurasian Pine Martens Martes martes during Cycles in Microtine Density.” Ecography 22 (3): 324–36.
Helldin, J. O. 2000. “Seasonal Diet of Pine Marten Martes martes in Southern Boreal Sweden.” Acta Theriologica 45 (September): 409–20.
Helldin, J. O. 2000. “Population Trends and Harvest Management of Pine Marten Martes martes in Scandinavia.” Wildlife Biology 6 (2): 111–20.
Heptner, V. G., and N. P. Naumov. 1998. Mammals of the Soviet Union. Sirenia and Carnivora (Sea Cows; Wolves and Bears). Vol. 2(1a). Washington, DC: Science Publishers.
Herr, J., L. Schley, and T. J. Roper. 2009. “Socio‐spatial Organization of Urban Stone Martens.” Journal of Zoology 277 (1): 54–62.
Herr, J., Schley, L., Engel, E., and T. J. Roper. 2010. “Den Preferences and Denning Behaviour in Urban Stone Martens (Martes foina).” Mammalian Biology = Zeitschrift Fur Saugetierkunde 75 (2): 138–45.
Herrero, J., A. Kranz, D. Skumatov, A. V. Abramov, T. Maran, and V. G. Monakhov. 2016. “Martes martes. In: The IUCN Red List of Threatened Species 2016: e.T12848A45199169.” IUCN. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T12848A45199169.en.
Howell, B., K. Aubry, P. Happe, K. Jenkins, R. Long, P. MacKay, and K. Moriarty. 2020. “Pacific Marten (Martes caurina) Distribution Study on the Olympic Peninsula, Washington. Final Interagency Special Status/Sensitive Species (ISSSSP) Report, November 1, 2018-September 30, 2019.”
Ingeman, Kurt E., Lily Z. Zhao, Christopher Wolf, David R. Williams, Amelia L. Ritger, William J. Ripple, Kai L. Kopecky, et al. 2022. “Glimmers of Hope in Large Carnivore Recoveries.” Scientific Reports 12 (1): 10005.
IUCN-CMP. 2016. “Version 2.0 of Threats and Actions Classification” Conservation Standards. https://www.conservationstandards.org/library-item/threats-and-actions-taxonomies/
Kang, Wanmo, Taeyoung Choi, Gowoon Kim, and Donggul Woo. 2023. “Habitat, Connectivity, and Roadkill of Korea’s Apex Predator, the Yellow-Throated Marten.” Wildlife Research 51, WR21185.
Knaus, Brian J., Richard Cronn, Aaron Liston, Kristine Pilgrim, and Michael K. Schwartz. 2011. “Mitochondrial Genome Sequences Illuminate Maternal Lineages of Conservation Concern in a Rare Carnivore.” BMC Ecology 11 (April): 10.
Kordosky, Jennifer R., Eric M. Gese, Craig M. Thompson, Patricia A. Terletzky, Kathryn L. Purcell, and Jon D. Schneiderman. 2021. “Landscape Use by Fishers (Pekania pennanti): Core Areas Differ in Habitat than the Entire Home Range.” Canadian Journal of Zoology 99 (4): 289–97.
Kordosky, Jennifer R., Eric M. Gese, Craig M. Thompson, Patricia A. Terletzky, Lorin A. Neuman-Lee, Jon D. Schneiderman, Kathryn L. Purcell, and Susannah S. French. 2021. “Landscape of Stress: Tree Mortality Influences Physiological Stress and Survival in a Native Mesocarnivore.” PloS One 16 (7): e0253604.
Krebs, John, Eric Lofroth, Jeffrey Copeland, Vivian Banci, Dorothy Cooley, Howard Golden, Audrey Magoun, Robert Mulders, and Brad Shults. 2004. “Synthesis Of Survival Rates And Causes Of Mortality In North American Wolverines.” Wildfire 68 (3): 493–502.
Krohner, Jessica M., Paul M. Lukacs, Robert Inman, Joel D. Sauder, Justin A. Gude, Cory Mosby, Jessica A. Coltrane, Rebecca A. Mowry, and Joshua J. Millspaugh. 2022. “Finding Fishers: Determining Fisher Occupancy in the Northern Rocky Mountains.” The Journal of Wildlife Management 86 (2). https://doi.org/10.1002/jwmg.22162.
Kumar, A., and K. Yoganand. 1999. “Distribution and Abundance of Small Carnivores in Nilgiri Biosphere Reserve, India.” In ENVIS Bulletin. Vol 2(2): Mustelids, Viverrids and Herpestids of India, edited by S. A. Hussain, 2(2):76–86. Dehradun, India: Wildlife Institute of India.
Kyle, C. J., J. F. Robitaille, and C. Strobeck. 2001. “Genetic Variation and Structure of Fisher (Martes pennanti) Populations across North America.” Molecular Ecology 10 (9): 2341–47.
LaPoint, Scott, Gallery, Paul, Martin Wikelski, and Roland Kays. 2013. “Animal Behavior, Cost-Based Corridor Models, and Real Corridors.” Landscape Ecology 28 (8): 1615–30.
Lee, Hwa-Jin, Oh-Sun Lee, Dong-Gul Woo, Han-Na Kim, Mark C. Wallace, and Yeong-Seok Jo. 2021. “Current Distribution and Habitat Models of the Yellow-Throated Marten, Martes flavigula, in South Korea.” Mammal Research 66 (3): 429–41.
Lewis, J. C., J. I. Ransom, T. Chestnut, D. O. Werntz, S. Black, D. Whiteside, J. L. Postigo, and A. Moehrenschlager. 2022. “Cascades Fisher Reintroduction Project: Final Project Report. Natural Resource Report NPS/PWR/NRR—2022/2418.” Fort Collins, Colorado: National Park Service. https://doi.org/10.36967/2293605
Lewis, Jeffrey C., Patti J. Happe, Kurt J. Jenkins, and David J. Manson. 2012. “Olympic Fisher Reintroduction Project: Progress Report 2008–2011.” Washington Department of Fish and Wildlife. https://wdfw.wa.gov/sites/default/files/publications/01393/wdfw01393.pdf.
Lindström, Erik R., Scott M. Brainerd, J. O. Helldin, and Kristian Overskaug. 1995. “Pine Marten — Red Fox Interactions: A Case of Intraguild Predation?” Annales Zoologici Fennici 32 (1): 123–30.
Litchfield, K. 2019. “Mission Pine Marten Community Engagement & Consultation Report.” Gloucester, UK: Gloucester Wildlife Trust.
Lofroth, E. C., C. M. Raley, J. M. Higley, R. L. Truex, J. S. Yaeger, J. C. Lewis, P. J. Happe, et al. 2010. “Conservation of Fishers (Martes pennanti) in South-Central British Columbia, Western Washington, Western Oregon, and California – Volume I: Conservation Assessment.” Denver, Colorado, USA: USDI Bureau of Land Management.
Lofroth, Eric C., Richard D. Weir, Larry R. Davis, and Ingebjorg Jean Hansen. 2023. “A Tale of Two Populations: Vital Rates of Fishers in British Columbia, Canada.” The Journal of Wildlife Management 87 (1). https://doi.org/10.1002/jwmg.22315.
Loughry, Steven C., Maggie D. Triska, Dorothy M. Fecske, and Thomas L. Serfass. 2012. “A Direct Comparison of Enclosed Track Plates and Remote Cameras in Detecting Fishers, Martes pennanti, in North Dakota.” The Canadian Field-Naturalist 126 (4): 281–87.
Lozano, Jorge, Agnieszka Olszańska, Zebensui Morales-Reyes, Antonio A. Castro, Aurelio F. Malo, Marcos Moleón, José A. Sánchez-Zapata, et al. 2019. “Human-Carnivore Relations: A Systematic Review.” Biological Conservation 237 (September): 480–92.
MacPherson, J., Croose E., D. Bavin, D. O’Mahony, J. P. Somper, and N. Buttriss. 2014. “Feasibility Assessment for Reinforcing Pine Marten Numbers in England and Wales.” Ledbury, UK: Vincent Wildlife Trust.
MacPherson, J., Wright, P., Schumaker, N., & Watkins, S. 2024. Use of multi-modelling methods to inform conservation and reintroductions of pine marten Martes martes in Britain. Stacks Journal: 24004. https://doi.org/10.60102/stacks-24004.
MacPherson, J., and P. Wright. 2021. “Long-Term Strategic Recovery Plan for Pine Martens in Britain, Technical Report.” Ledbury, UK.: Vincent Wildlife Trust.
Magoun, Audrey J., Cristina R. Laird, Mark A. Keech, Patrick Valkenburg, Lincoln S. Parrett, and Martin D. Robards. 2018. “Predation on Caribou (Rangifer tarandus) by Wolverines (Gulo gulo) after Long Pursuits.” The Canadian Field-Naturalist 132 (4): 382–85.
Magoun, Audrey J., and Jeffrey P. Copeland. 1998. “Characteristics of Wolverine Reproductive Den Sites.” The Journal of Wildlife Management 62 (4): 1313–20.
Mallick, Jayanta. 2015. “Natural History, Distribution and Status of Himalayan Yellow-Throated Marten (Martes flavigula) in Northern West Bengal, India.” In Animal Diversity Natural History and Conservation, edited by V.K. Gupta and A.K. Verma, 4:363–94. Daya Publishing House.
Manlick, Philip J., James E. Woodford, Benjamin Zuckerberg, and Jonathan N. Pauli. 2017. “Niche Compression Intensifies Competition between Reintroduced American Martens (Martes americana) and Fishers (Pekania pennanti).” Journal of Mammalogy 98 (3): 690–702.
Manlick, Philip J., Steve K. Windels, James E. Woodford, and Jonathan N. Pauli. 2020. “Can Landscape Heterogeneity Promote Carnivore Coexistence in Human-Dominated Landscapes?” Landscape Ecology 35 (9): 2013–27.
Marneweck, C., A. R. Butler, L. C. Gigliotti, S. N. Harris, A. J. Jensen, M. Muthersbaugh, B. A. Newman, et al. 2021. “Shining the Spotlight on Small Mammalian Carnivores: Global Status and Threats.” Biological Conservation 255 (March): 109005.
Marneweck, Courtney J., Benjamin L. Allen, Andrew R. Butler, Emmanuel Do Linh San, Stephen N. Harris, Alex J. Jensen, Elizabeth A. Saldo, et al. 2022. “Middle‐out Ecology: Small Carnivores as Sentinels of Global Change.” Mammal Review 52 (4): 471–79.
Martin, Marie E., Katie M. Moriarty, and Jonathan N. Pauli. 2020. “Forest Structure and Snow Depth Alter the Movement Patterns and Subsequent Expenditures of a Forest Carnivore, the Pacific Marten.” Oikos 129 (3): 356–66.
Mason, Natalie, Michelle Ward, James E. M. Watson, Oscar Venter, and Rebecca K. Runting. 2020. “Global Opportunities and Challenges for Transboundary Conservation.” Nature Ecology & Evolution 4 (5): 694–701.
Mathews, F., Kubasiewicz, L., Gurnell, J., Harrower, C., McDonald, R. & Shore, R. 2018. “A review of the population and conservation status of British mammals.” A report by the Mammal Society under contract to Natural England, Natural Resources Wales and Scottish Natural Heritage. Natural England, Peterborough.
Maxwell, Sean L., Richard A. Fuller, Thomas M. Brooks, and James E. M. Watson. 2016. “Biodiversity: The Ravages of Guns, Nets and Bulldozers.” Nature 536 (7615): 143–45.
May, Roel. 09 2007. “Spatial Ecology of Wolverines in Scandinavia.” Edited by Arild Landa (NINA) and Reidar Andersen (NTNU). Doctor of Philosophy, Norwegian University of Science and Technology. https://doi.org/10.13140/RG.2.1.1647.8489.
McDonald, Robert I., and Timothy M. Boucher. 2011. “Global Development and the Future of the Protected Area Strategy.” Biological Conservation 144 (1): 383–92.
McKelvey, Kevin S., Jeffrey P. Copeland, Michael K. Schwartz, Jeremy S. Littell, Keith B. Aubry, John R. Squires, Sean A. Parks, Marketa M. Elsner, and Guillaume S. Mauger. 2011. “Climate Change Predicted to Shift Wolverine Distributions, Connectivity, and Dispersal Corridors.” Ecological Applications: A Publication of the Ecological Society of America 21 (8): 2882–97.
Monakhov, V. G. 2011. “Martes zibellina (Carnivora: Mustelidae).” Mammalian Species 43 (1): 75–86.
Monakhov, V. G. 2015. “Mass Reintroduction of Sable (Martes zibellina): Achievements and Problems over 55 Years.” Martes Working Group Newsletter 21: 25–33.
Monakhov, V. G. 2022. “Martes martes (Carnivora: Mustelidae).” Mammalian Species 54 (1022): seac007.
Moore, Patrick, and Angela Wheelock. 1990. Wolverine Myths and Visions: Dene Traditions from Northern Alberta. U of Nebraska Press.
Moqanaki, Ehsan, Cyril Milleret, Pierre Dupont, Henrik Brøseth, and Richard Bischof. 2023. “Wolverine Density Distribution Reflects Past Persecution and Current Management in Scandinavia.” Ecography, June. https://doi.org/10.1111/ecog.06689.
Moriarty, K. M., J. Thompson, M. Delheimer, B. R. Barry, M. Linnell, T. Levi, K. Hamm, D. Early, H. Gamblin, M. S. Gunther, J. Ellison, J. S. Prevey, J. Hartman, and R.Davis. 2021. “Predicted Distribution of a Rare and Understudied Forest Carnivore: Humboldt Marten (Martes caurina humboldtensis).” PeerJ 9: e11670.
Moriarty, K. M., K. A. Aubry, C. N. Morozumi, B. L. Howell, P. J. Happe, K. J. Jenkins, K. L. Pilgrim, and M. K. Schwartz. 2019. “Status of Pacific Martens (Martes caurina) on the Olympic Peninsula, Washington.” Northwest Science: Official Publication of the Northwest Scientific Association 93 (2): 122–36.
Moriarty, Katie M., Clinton W. Epps, Matthew G. Betts, Dalton J. Hance, J. D. Bailey, and William J. Zielinski. 2015. “Experimental Evidence That Simplified Forest Structure Interacts with Snow Cover to Influence Functional Connectivity for Pacific Martens.” Landscape Ecology 30 (10): 1865–77.
Mowat, Garth, Anthony P. Clevenger, Andrea D. Kortello, Doris Hausleitner, Mirjam Barrueto, Laura Smit, Clayton Lamb, Benjamin DorsEy, and Peter K. Ott. 2020. “The Sustainability of Wolverine Trapping Mortality in Southern Canada.” The Journal of Wildlife Management 84 (2): 213–26.
Mudappa, D., D. Jathana, and T. R. S. Raman. 2015. “Martes gwatkinsii. The IUCN Red List of Threatened Species 2015: e.T12847A45199025.” IUCN. https://doi.org/10.2305/IUCN.UK.2015-.
Naney, R. H., L. L. Finley, E. C. Lofroth, P. J. Happe, A. L. Krause, C. M. Raley, R. L. Truex, et al. 2012. “Conservation of Fishers (Martes pennanti) in South-Central British Columbia, Western Washington, Western Oregon, and California–Volume III: Threat Assessment.” Denver, Colorado, USA: USDI Bureau of Land Management. https://a100.gov.bc.ca/pub/acat/documents/r25101/ConservFisherVol3_1331594845977_f4920e8f3b404c8943a9753022f757e7bebe50c514b60a474b6ccd1327a41335.pdf.
Oleynikov, A. Yu, V. G. Yudin, G. P. Salkina, and G. A. Sedash. 2022. “Current Population Status of Yellow-Throated Marten Martes (Charronia) flavigula in Russia.” Russian Journal of Theriology 21 (1): 63–69.
Oleynikov, Yu A., and K. N. Tkachenko. 2013. “The Necessity of Including the Bengal Cat and the Yellow-Throated Marten in the Red Book of Russia.” In Preservation of Animal Diversity and the Hunting Economy of Russia, edited by G. I. Blokhin, 312–14. Moscow: Moscow Timiryazev Agricultural Academy.
O’Brien, Patrick, Chris Bernier, and Paul Hapeman. 2018. “A New Record of an American Marten (Martes americana) Population in Southern Vermont.” Small Carnivore Conservation: The Newsletter and Journal of the IUCN/SSC Mustelid, Viverrid & Procynid Specialist Group 56: 68–75.
Papakosta, Malamati, Kyriaki Kitikidou, Dimitris Bakaloudis, and Christos Vlachos. 2014. “Dietary Variation of the Stone Marten (Martes foina): A Meta-Analysis Approach.” Wildlife Biology in Practice 10 (2). https://doi.org/10.2461/wbp.2014.10.11.
Parisien, Marc-André, Quinn E. Barber, Mathieu L. Bourbonnais, Lori D. Daniels, Mike D. Flannigan, Robert W. Gray, Kira M. Hoffman, et al. 2023. “Abrupt, Climate-Induced Increase in Wildfires in British Columbia since the Mid-2000s.” Communications Earth & Environment 4 (1): 1–11.
Parr, John, and Will Duckworth. 04 2007. “Notes on Diet, Habituation and Sociality of Yellow-Throated Martens Martes flavigula.” Small Carnivore Conservation: The Newsletter and Journal of the IUCN/SSC Mustelid, Viverrid & Procynid Specialist Group 36.
Pauli, Jonathan N., Benjamin Zuckerberg, John P. Whiteman, and Warren Porter. 2013. “The Subnivium: A Deteriorating Seasonal Refugium.” Frontiers in Ecology and the Environment 11 (5): 260–67.
Pauli, Jonathan N., Philip J. Manlick, Jody M. Tucker, G. Bradley Smith, Paul G. Jensen, and Jason T. Fisher. 2022. “Competitive Overlap between Martens Martes americana and Martes caurina and Fishers Pekania pennanti: A Rangewide Perspective and Synthesis.” Mammal Review 52 (3): 392–409.
Peeva, S., and E. Raichev. 2016. “Stone Marten (Martes foina, Erxl., 1777) and Villagers: Human-Wildlife Social Conflict.” Agricultural Science and Technology 8: 158–61.
Pereira, Lúcia, Ana Vasques, Paula Maia, Maria João Ramos Pereira, Carlos Fonseca, and Milena Matos. 2019. “Native and Exotic Seed Dispersal by the Stone Marten (Martes foina): Implications for the Regeneration of a Relict Climactic Forest in Central Portugal.” Integrative Zoology 14 (3): 280–92.
Pocock, R. I. 1941. The Fauna of British India, Including Ceylon and Burma. Mammalia. Vol. 2. London: Taylor and Francis.
Powell, R. A., J. C. Lewis, B. G. Slough, S. M. Brainerd, N. R. Jordan, A. V. Abramov, V. Monakhov, P. A. Zollner, and T. Murakami. 2012. “Evaluating Translocations of Martens, Sables, and Fishers: Testing Model Predictions with Field Data.” In Biology and Conservation of Martens, Sables, and Fishers: A New Synthesis., edited by K. B. Aubry, W. J. Zielinski, M. G. Raphael, G. Proulx, and S. W. Buskirk, 93–137. Ithaca, New York, USA: Cornell University Press.
Powell, R. A., S. W. Buskirk, and W. J. Zielinski. 2003. “Fisher and Marten.” In Wild Mammals of North America: Biology, Management, and Conservation, edited by G. A. Feldhamer, 635–49). Baltimore, MD: The John Hopkins University Press.
Powell, Roger A., and I. D. Thompson. 1993. “Why Do Some Forest Carnivores Exhibit Intrasexual Territoriality and What Are the Consequences for Management.” Proc Int Union Game Biol 21: 268–73.
Presley, Steven J. 2000. “Eira barbara.” Mammalian Species 2000 (636): 1–6.
Proulx, Gilbert, and Keith B. Aubry. 2020. “The Martes Complex: Opportunities for Developing Multi-Species Management and Conservation Strategies.” Canadian Wildlife Biology & Management 9 (2): 49–67.
Ripple, William J., James A. Estes, Robert L. Beschta, Christopher C. Wilmers, Euan G. Ritchie, Mark Hebblewhite, Joel Berger, et al. 2014. “Status and Ecological Effects of the World’s Largest Carnivores.” Science 343 (6167): 1241484.
Ruggiero, Leonard F., Keith B. Aubry, Steven W. Buskirk, L. Jack Lyon, and William J. Zielinski. 1994. “The Scientific Basis for Conserving Forest Carnivores: American Marten, Fisher, Lynx, and Wolverine in the Western United States.” Ft. Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station. https://doi.org/10.2737/rm-gtr-254.
Sainsbury, Katherine A., Richard F. Shore, Henry Schofield, Elizabeth Croose, Ruairidh D. Campbell, and Robbie A. Mcdonald. 2019. “Recent History, Current Status, Conservation and Management of Native Mammalian Carnivore Species in Great Britain.” Mammal Review 49 (2): 171–88.
Schiaffini, Mauro I., Francisco J. Prevosti, Brenda S. Ferrero, and Jorge I. Noriega. 2017. “A Late Pleistocene Guloninae (Carnivora, Mustelidae) from South America (Argentina, Entre Ríos Province), Biogeographic Implications.” Journal of South American Earth Sciences 78 (October): 141–49.
Schiaffini, Mauro Ignacio. 2020. “Are Subspecies (of Eira barbara) Real?” Journal of Mammalogy 101 (5): 1410–25.
Schreiber, A., R. Wirth, M. Riffel, and H. V. Rompaey. 1989. “Weasels, Civets, Mongooses, and Their Relatives: An Action Plan for the Conservation of Mustelids and Viverrids.” IUCN. https://policycommons.net/artifacts/1374093/weasels-civets-mongooses-and-their-relatives/1988330/.
Schwartz, Michael K., Ashley D. Walters, Kristine L. Pilgrim, Katie M. Moriarty, Keith M. Slauson, William J. Zielinski, Keith B. Aubry, et al. 2020. “Pliocene-Early Pleistocene Geological Events Structure Pacific Martens (Martes caurina).” The Journal of Heredity 111 (2): 169–81.
Scrafford, Matthew A., Jacob L. Seguin, Laura K. McCaw, Mark S. Boyce, and Justina C. Ray. 2024. “Wolverine Density, Survival, and Population Trends in the Canadian Boreal Forest.” The Journal of Wildlife Management 88(5), e22587. https://doi.org/10.1002/jwmg.22587.
Shameer, Thekke Thumbath, Ninad Avinash Mungi, Sulekha Jameela Backer, Sreehari Raman, Srinivas Ramchandra Reddy, Pulinkunel Sayedmohammed Easa, and Raveendranathanpillai Sanil. 2023. “Distribution and Conservation Status of the Endemic Nilgiri Marten (Martes gwatkinsii).” Mammalia 87 (4): 360–66.
Smith, Felisa A., S. Kathleen Lyons, S. K. Morgan Ernest, Kate E. Jones, Dawn M. Kaufman, Tamar Dayan, Pablo A. Marquet, James H. Brown, and John P. Haskell. 2003. “Body Mass of Late Quaternary Mammals.” Ecology 84 (12): 3403–3403.
Smith, Matthew M., John D. Erb, and Jonathan N. Pauli. 2023. “Reciprocated Competition between Two Forest Carnivores Drives Dietary Specialization.” The Journal of Animal Ecology 92 (9): 1695–1706.
Sojuzpushnina. 2024. “Sales Results of 226 International Fur Auction.” Sojuzpushnina. May 21, 2024. https://sojuzpushnina.ru/en/news/detail?id=262.
Soley, Fernando G., and Isaías Alvarado-Díaz. 2011. “Prospective Thinking in a Mustelid? Eira barbara (Carnivora) Cache Unripe Fruits to Consume Them Once Ripened.” Die Naturwissenschaften 98 (8): 693–98.
Spencer, Wayne D., Heather Rustigian-Romsos, Ken Ferschweiler, and Dominique Bachelet. 2015. “Simulating Effects of Climate and Vegetation Change on Distributions of Martens and Fishers in the Sierra Nevada, California, Using Maxent and MC1.” In Global Vegetation Dynamics, 135–49. Hoboken, NJ: John Wiley & Sons, Inc.
Steel, Zachary L., Hugh D. Safford, and Joshua H. Viers. 2015. “The Fire Frequency-Severity Relationship and the Legacy of Fire Suppression in California Forests.” Ecosphere 6 (1): art8.
Steventon, J. Douglas, and David K. Daust. 2009. “Management Strategies for a Large-Scale Mountain Pine Beetle Outbreak: Modelling Impacts on American Martens.” Forest Ecology and Management 257 (9): 1976–85.
Stewart, F. E. C., J. P. Volpe, B. R. Eaton, and G. A. Hood. 2019. “Protected Areas Alone Rarely Predict Mammalian Biodiversity across Spatial Scales in an Albertan Working Landscape.” Biologicals: Journal of the International Association of Biological Standardization. https://www.sciencedirect.com/science/article/pii/S0006320719308584.
Stewart, Frances E. C., John P. Volpe, John S. Taylor, Jeff Bowman, Philippe J. Thomas, Margo J. Pybus, and Jason T. Fisher. 2017. “Distinguishing Reintroduction from Recolonization with Genetic Testing.” Biological Conservation 214 (October): 242–49.
Streicher, Jarryd P., Tharmalingam Ramesh, and Colleen T. Downs. 2023. “Not All Mammalian Small Carnivores Are Equal: A Global Review of the Research Effort in Urban Areas.” African Journal of Wildlife Research 53 (1). https://doi.org/10.3957/056.053.0072.
Suffice, P., Asselin, H., Imbeau, L., Cheveau, M., and P. Drapeau. 2017. “More Fishers and Fewer Martens due to Cumulative Effects of Forest Management and Climate Change as Evidenced from Local Knowledge.” Journal of Ethnobiology and Ethnomedicine 13 (1): 51.
Suffice, P., Cheveau, M., Imbeau, L., Mazerolle, M. J., Asselin, H., and P. Drapeau. 2020. “Habitat, Climate, and Fisher and Marten Distributions.” The Journal of Wildlife Management 84 (2): 277–92.
Swenson, J., and Henrik Andrén. 2005. “People and Wildlife: A Tale of Two Countries: Large Carnivore Depredation and Compensation Schemes in Sweden and Norway.” https://doi.org/10.1017/CBO9780511614774.021.
Thompson, Kimberly L., Benjamin Zuckerberg, Warren P. Porter, and Jonathan N. Pauli. 2021. “The Decline of a Hidden and Expansive Microhabitat: The Subnivium.” Frontiers in Ecology and the Environment 19 (5): 268–73.
Tsuji, Yamato, Risma Yanti, Atsushi Takizawa, and Toshio Hagiwara. 2020. “Interspecific Difference in Seed Dispersal Characteristics between Japanese Macaques (Macaca fuscata) and Sympatric Japanese Martens (Martes melampus).” Folia Primatologica; International Journal of Primatology 91 (6): 711–20.
Tucker, Jody M., Michael K. Schwartz, Richard L. Truex, Samantha M. Wisely, and Fred W. Allendorf. 2014. “Sampling Affects the Detection of Genetic Subdivision and Conservation Implications for Fisher in the Sierra Nevada.” Conservation Genetics 15 (1): 123–36.
Walker, C. W., C. Vilà, A. Landa, M. Lindén, and H. Ellegren. 2001. “Genetic Variation and Population Structure in Scandinavian Wolverine (Gulo gulo) Populations.” Molecular Ecology 10 (1): 53–63.
Weir, Richard D., Andrew M. Rankin, Lacy Robinson, Kristine L. Pilgrim, Michael K. Schwartz, and Michael K. Lucid. 2024. “Genetic Structuring of Fishers in British Columbia, Canada: Implications for Population Conservation and Management.” Journal of Mammalogy 105 (3): 465-480.
Weir, Richard D., and Fraser B. Corbould. 2010. “Factors Affecting Landscape Occupancy by Fishers in North‐central British Columbia.” The Journal of Wildlife Management 74 (3): 405–10.
Wereszczuk, Anna, Tim R. Hofmeester, Alexander Csanády, Tomislav Dumić, Morten Elmeros, József Lanszki, Aksel B. Madsen, et al. 2021. “Different Increase Rate in Body Mass of Two Marten Species due to Climate Warming Potentially Reinforces Interspecific Competition.” Scientific Reports 11 (1): 24164.
Wereszczuk, Anna, and Andrzej Zalewski. 2019. “Does the Matrix Matter? Home Range Sizes and Space Use Strategies in Stone Marten at Sites with Differing Degrees of Isolation.” Mammal Research 64 (1): 71–85.
Wereszczuk, Anna, and Andrzej Zalewski. 2023. “An Anthropogenic Landscape Reduces the Influence of Climate Conditions and Moonlight on Carnivore Activity.” Behavioral Ecology and Sociobiology 77 (5): 55.
Wisely, Samantha M., Steven W. Buskirk, Gregory A. Russell, Keith B. Aubry, and William J. Zielinski. 2004. “Genetic Diversity and Structure of the Fisher (Martes pennanti) in a Peninsular and Peripheral Metapopulation.” Journal of Mammalogy 85 (4): 640–48.
Woollard, Tyler F., Daniel J. Harrison, Erin M. Simons-Legaard, and Kirstin E. Fagan. 2024. “Functional Responses in American Marten Habitat Selection Indicate Cumulative Effects of Progressive Habitat Change.” Ecosphere 15 (1).
Wright, Philip L. 1953. “Intergradation between Martes americana and Martes caurina in Western Montana.” Journal of Mammalogy 34 (1): 74–86.
Yudin, V. G. 2022. “On the Need to Introduce the Yellow-Throated Marten, Martes flavigula Boddaert, 1785 to the Red Data Book of Primorsky Krai.” Biota and Environment of Natural Areas 10 (2): 37–47.
Yudin, V. G., and E. V. Yudina. 2022. “The Yellow-Throated Marten of the Far East of Russia.”
Zielinski, William J., Keith M. Slauson, Carlos R. Carroll, Christopher J. Kent, and Donald G. Kudrna. 2001. “Status of American Martens in Coastal Forests of the Pacific States.” Journal of Mammalogy 82 (2): 478–90.
Open Access
Peer-Reviewed
Creative Commons
Submitted: 15 June 2024
Accepted: 27 March 2025
Published: 14 August 2025
The authors were supported by their respective institutions while writing this manuscript.
Conflicts of Interest:
The authors declare no conflicts of interest.